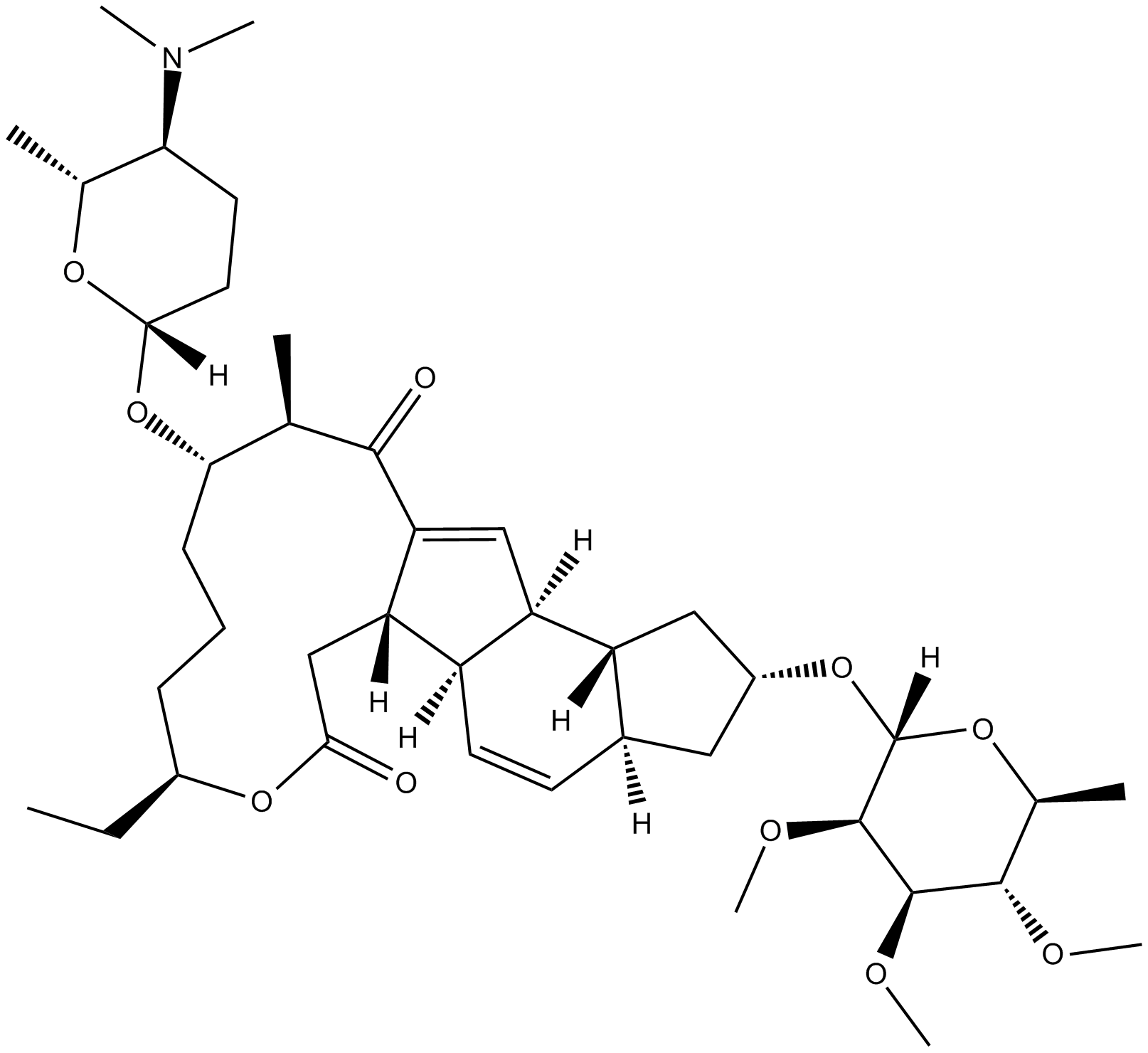
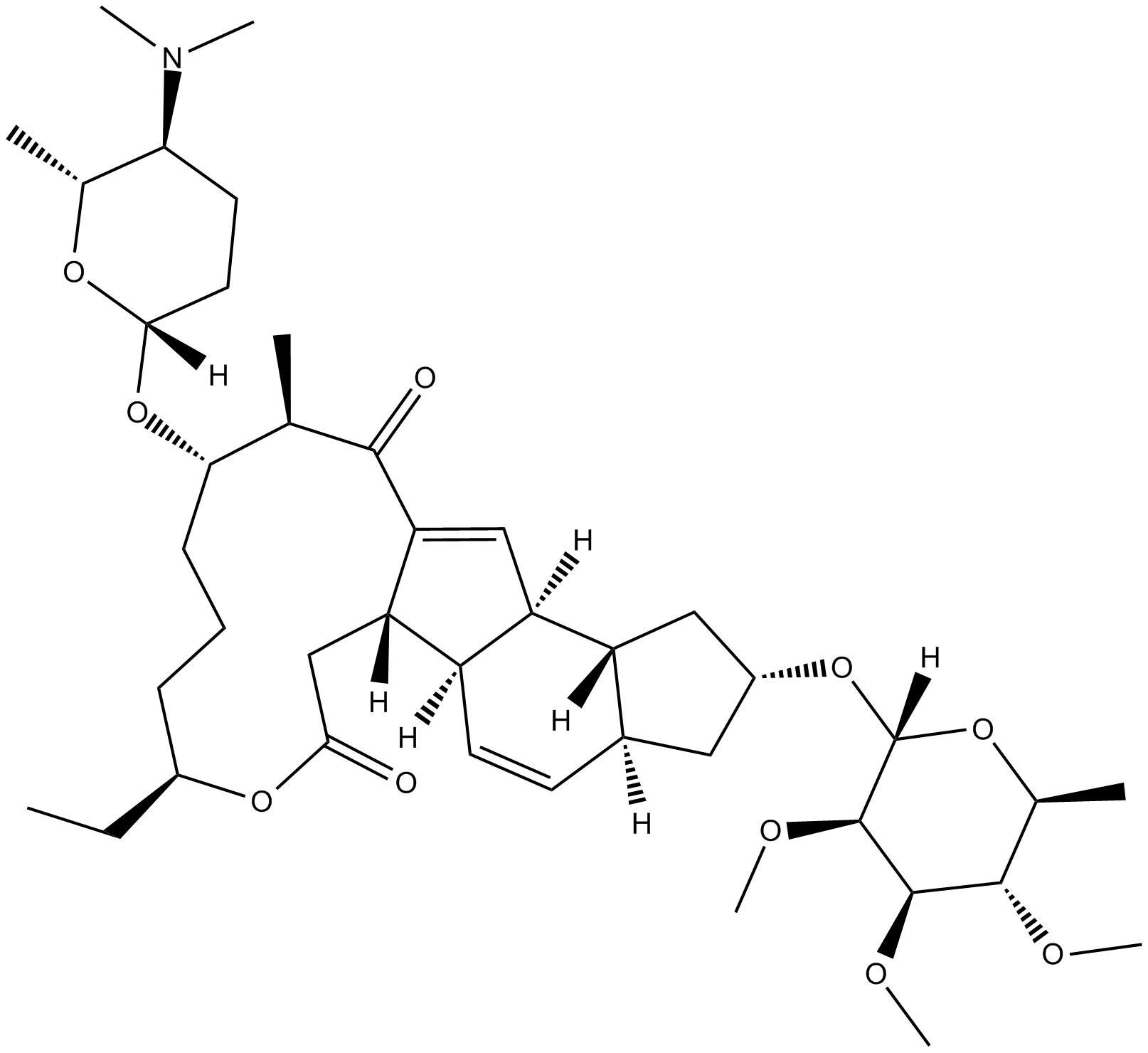
Spinosyn A
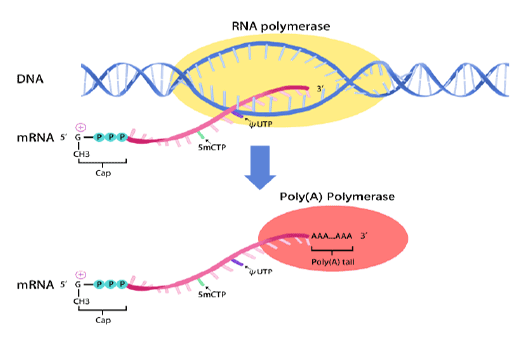
mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail
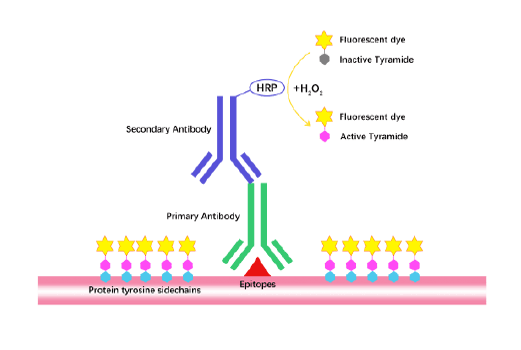
Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.
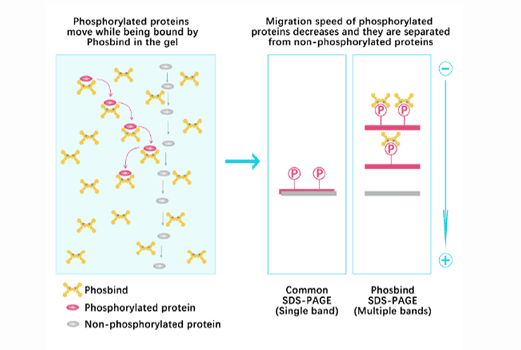
Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody
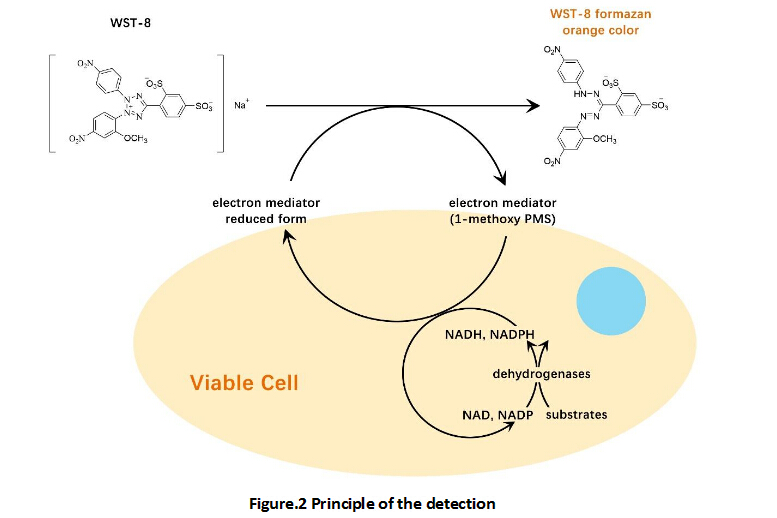
Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay
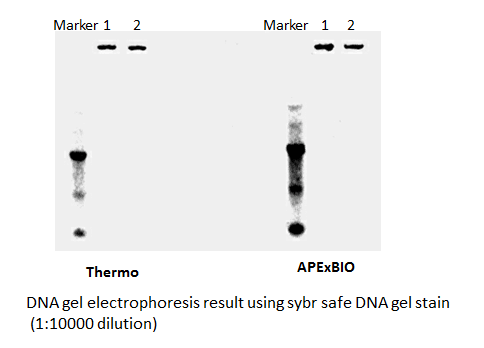
SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.
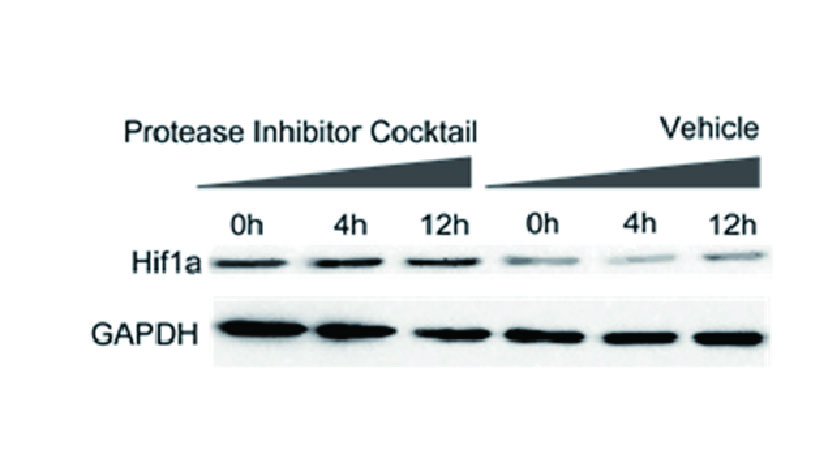
Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Spinosyn A is an insect nicotinic acetylcholinesterase receptors (nAChRs) agonist and potent insecticide.
The spinosyns are a family of macrolide natural products produced by the soil microorganism Saccharopolyspora spinosa. Spinosyn A is identified as a naturally-occurring macrocyclic lactone that is a potent insecticide.
In vitro: The mixture of spinosyns A and D, a commercial insecticide TracerTM (DowAgroSciences), is useful against various crop pests such as tobacco budworm. It was found that the deoxy analogs of spinosyns A were more potent insecticides than their respective parent factor. Moreover, the 2’-desmethoxy analogs of spinosyns A showed insecticidal potency against H. virescens greater than that of spinosyns A and D, suggesting that polarity was not well tolerated. Furthermore, the activity of 3'-deoxy spinosyn J was about the same as spinosyn A, and the activity of 2'-deoxy spinosyn H was found to be slightly greater than that of spinosyn A [1].
In vivo: Currently, there is no animal in vivo data reported.
Clinical trial: So far, no clinical study has been conducted.
Reference:
[1] L. C. Creemer, H. A. Kirst, J. W. Paschal, et al. Synthesis and insecticidal activity of spinosyn analogs functionally altered at the 2'-,3'- and 4'-positions of the rhamnose moiety. J.Antibiot.(Tokyo) 53(2), 171-178 (2000).
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 732 |
| Cas No. | 131929-60-7 |
| Formula | C41H65NO10 |
| Synonyms | A-83543A|Lepicidin A |
| Solubility | Soluble in DMSO |
| Chemical Name | (2R,3aS,5aR,5bS,9S,13S,14R,16aS,16bR)-2-[(6-deoxy-2,3,4-tri-O-methyl-α-L-mannopyranosyl)oxy]-13-[[(2R,5S,6R)-5-(dimethylamino)tetrahydro-6-methyl-2H-pyran-2-yl]oxy]-9-ethyl-2,3,3a,5a,5b,6,9,10,11,12,13,14,16a,16b-tetradecahydro-14-methyl-1H-as-indaceno[3, |
| SDF | Download SDF |
| Canonical SMILES | CN(C)[C@H]1CC[C@@](O[C@H]([C@@H](C)C2=O)CCC[C@H](CC)OC(C[C@]3([H])C2=C[C@]4([H])[C@@]3([H])C=C[C@@]5([H])[C@@]4([H])C[C@H](O[C@]6([H])O[C@@H](C)[C@H](OC)[C@@H](OC)[C@H]6OC)C5)=O)([H])O[C@@H]1C |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构