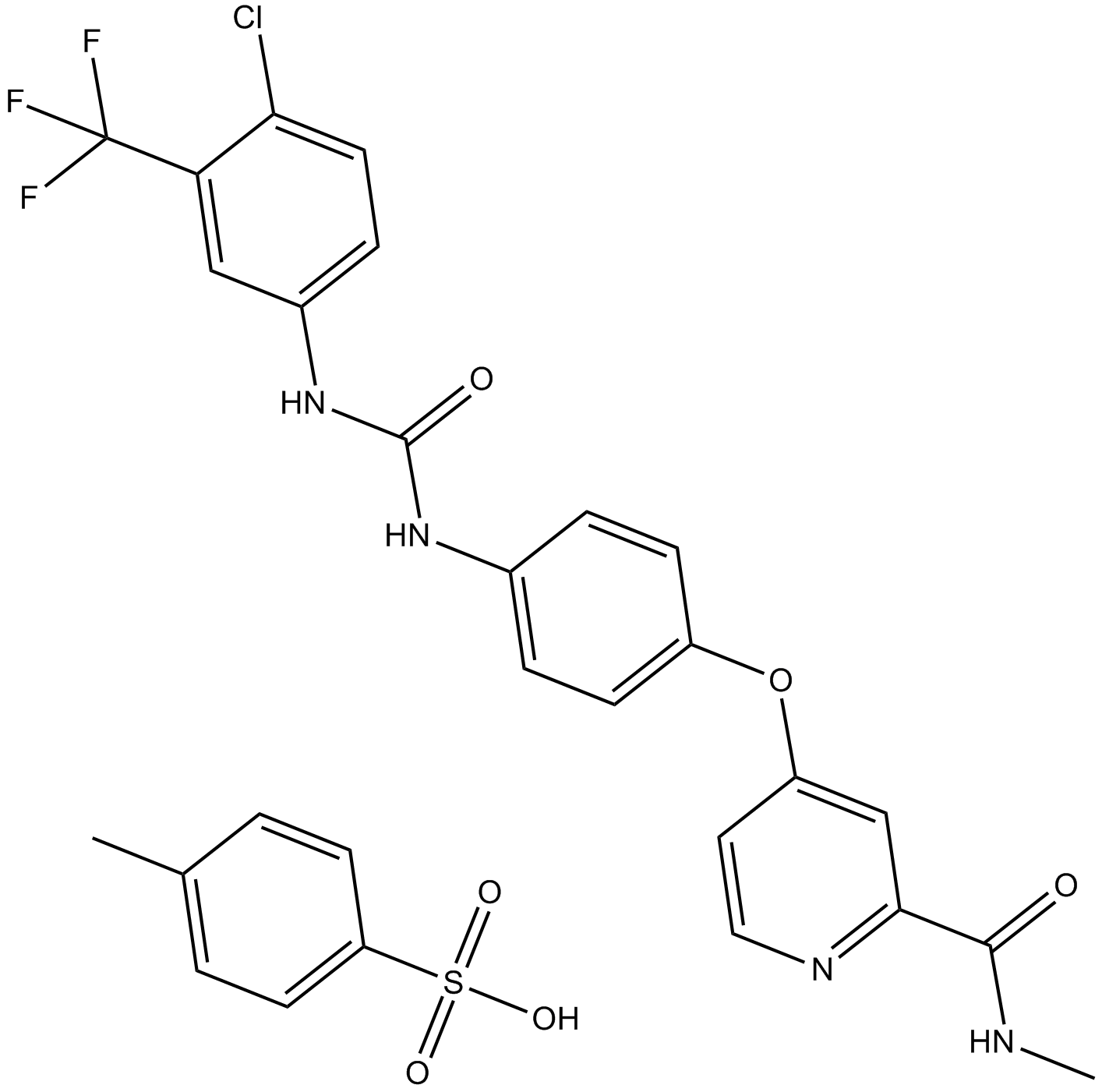
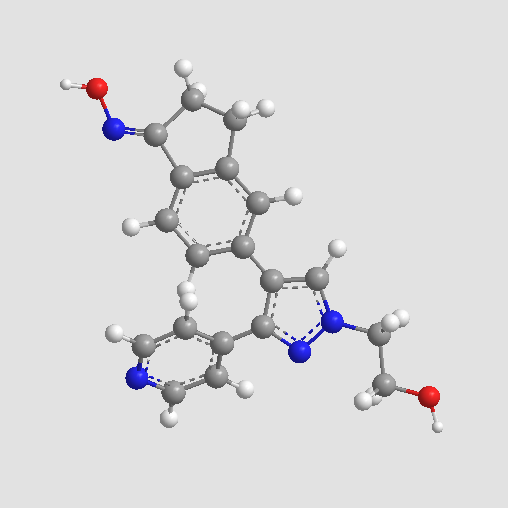
Sorafenib Tosylate
mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail
Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.
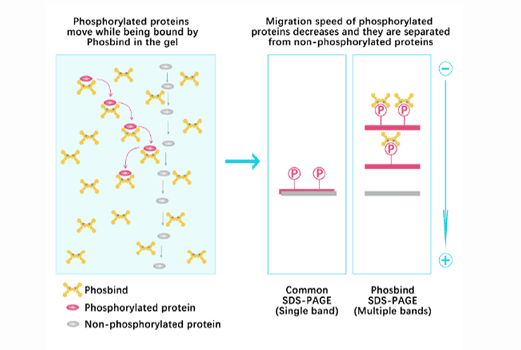
Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody
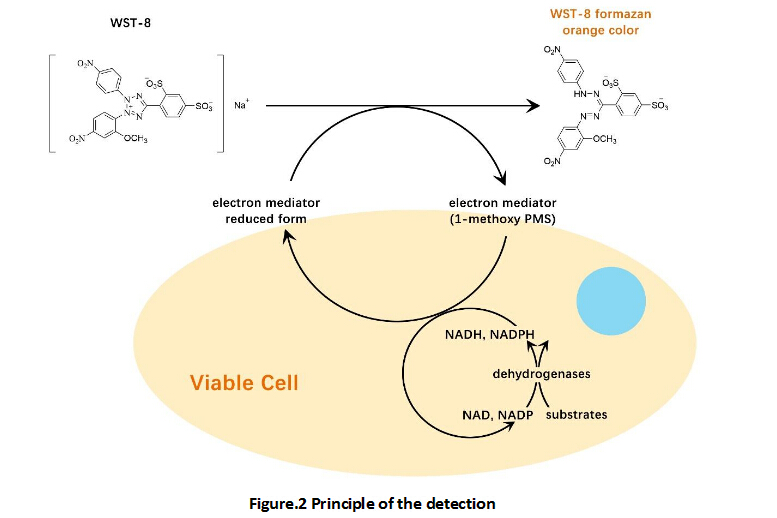
Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay
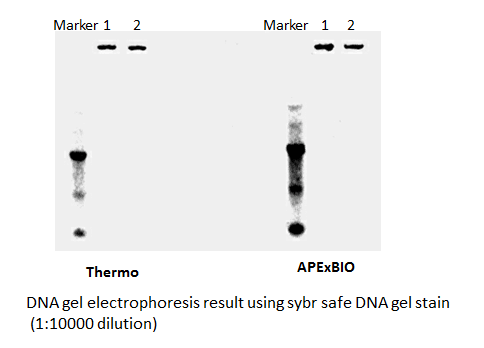
SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.
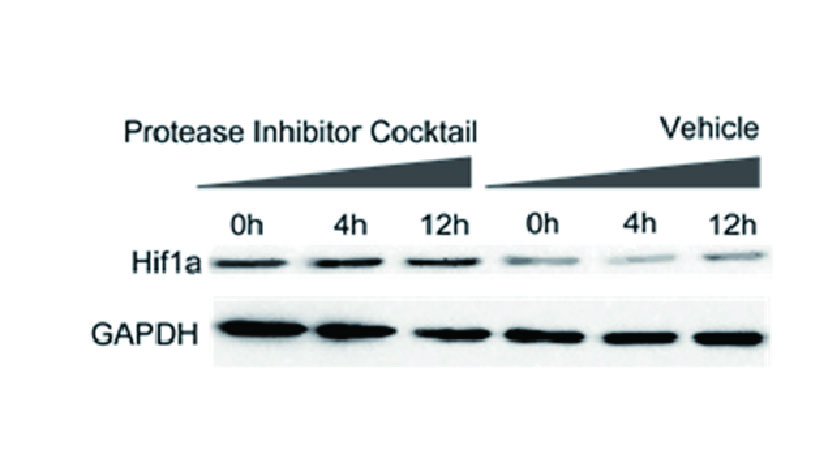
Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Sorafenib tosylate(甲苯磺酸索拉非尼),也称为Nexavar(多吉美),是一种小分子抗癌药物[1],也是一种新型可口服的Raf激酶和血管内皮生长因子受体(VEGFR)抑制剂,抑制肿瘤细胞的增殖和肿瘤血管生成[2]。在HepG2细胞(1×106)中,sorafenib tosylate的IC50值为2.09 μg/ml[3]。
Raf是一种有丝分裂原刺激蛋白激酶,作为信号级联反应的成分,刺激有丝分裂原活化蛋白激酶[4]。血管内皮生长因子(VEGF)是血管内皮细胞高特异性的有丝分裂原[5]。
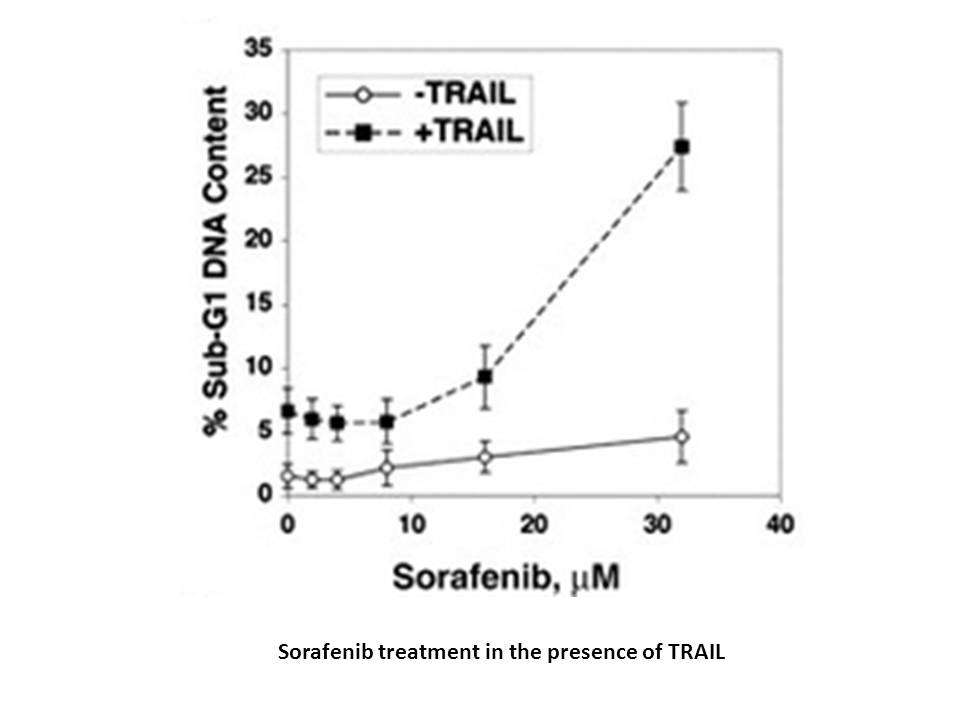
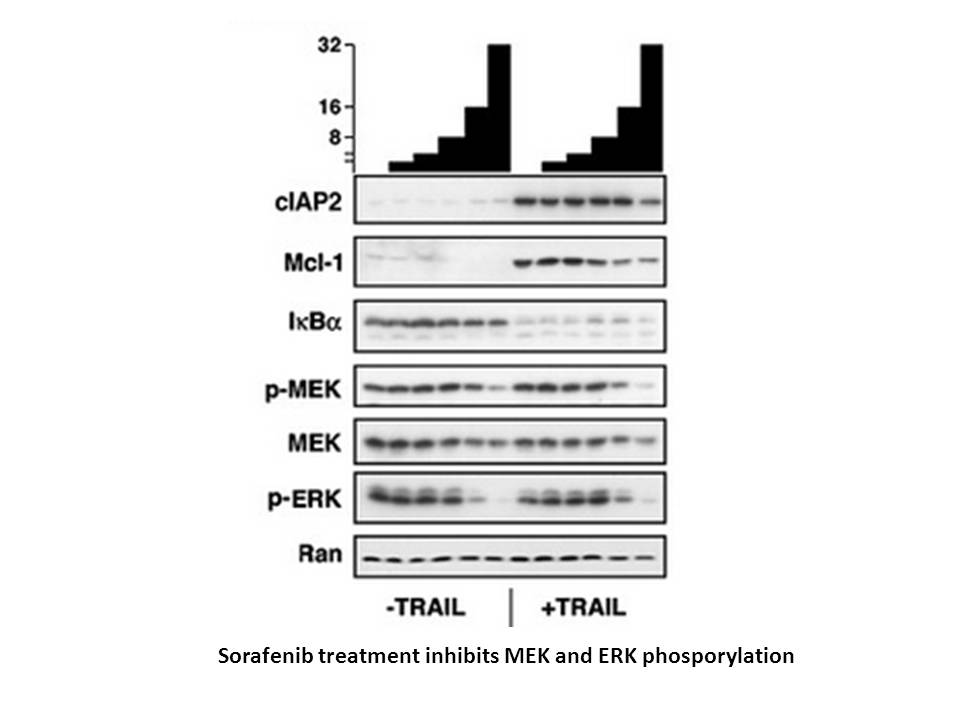
在MV4-11(FLT3-ITD)细胞中,nexavar以剂量依赖的方式有效抑制细胞增殖,IC50值为0.88 nM。在MV4-11细胞中,sorafenib tosylate在100 nM浓度时诱导43.6±5.2%的细胞凋亡,而在EOL-1细胞中,在浓度低至10 nM时可诱导89.29±1.8%的细胞凋亡[6]。
在注射105个MDA-MB-231细胞的6周龄裸大鼠中,与对照组相比,sorafenib tosylate单独给药后,在第45天和第55天观察到溶骨性病变体积的显著减少,在第55天观察到软组织成分的显著减少(p < 0.05)。与对照相比,sorafenib tosylate治疗使得骨转移的振幅A显著下降,从第35天一直持续到第55天(振幅A:p<0.01;维持,第35天和第55天,p<0.01;第45天p<0.05)[7]。
参考文献:
[1]. Chetan Lathia, John Lettieri, Frank Cihon, et al. Lack of effect of ketoconazole-mediated CYP3A inhibition on sorafenib clinical pharmacokinetics. Cancer Chemother Pharmacol, 2006, 57: 685-692.
[2]. M. J. Gnoth, S. Sandmann, K. Engel, et al. In Vitro to In Vivo Comparison of the Substrate Characteristics of Sorafenib Tosylate toward P-Glycoprotein. Drug Metabolism & Disposition, 2010, 38: 1341–1346.
[3]. Sayantan Dey, Subhadeep Roy, Nilanjana Deb, et al. Anti-carcinogenic Activity of Ruellia Tuberosa L. (Acanthaceae) Leaf Extract on Hepatoma Cell Line & Increased Superoxide Dismutase Activity on Macrophage Cell Lysate. Int J Pharm Pharm Sci, 2010, 5(Suppl 3): 854-861.
[4]. Markus Wartmann and Roger J. Davis. The Native Structure of the Activated Raf Protein Kinase Is a Membrane-bound Multi-subunit Complex. The Journal of Biological Chemistry, 1994, 269(9): 6695-6701.
[5]. Gera Neufeld, Tzafra Cohen, Stela Gengrinovitch, et al. Vascular endothelial growth factor (VEGF) and its receptors. The FASEB Journal, 1999, 13: 9-22.
[6]. D Auclair, D Miller, V Yatsula, et al. Antitumor activity of sorafenib in FLT3-driven leukemic cells. Leukemia, 2007, 21:439-445.
[7]. Maximilian Merz, Dorde Komljenovic, Stefan Zwick, et al. Sorafenib tosylate and paclitaxel induce anti-angiogenic, anti-tumour and anti-resorptive effects in experimental breast cancer bone metastases. European Journal of Cancer, 2011, 47:277-286.
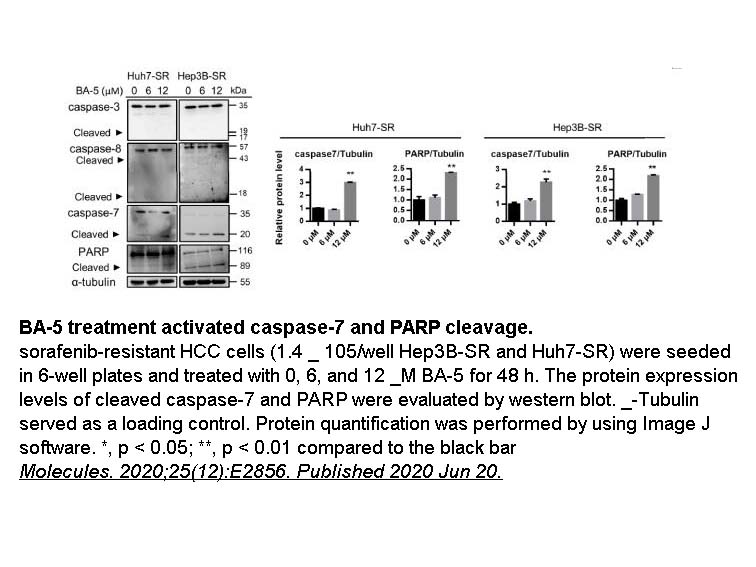
- 1. Liao YJ, Hsu SM, et al. "Treatment with a New Barbituric Acid Derivative Exerts Antiproliferative and Antimigratory Effects against Sorafenib Resistance in Hepatocellular Carcinoma." Molecules. 2020;25(12):E2856. PMID:32575795
- 2. Suk FM, Liu CL, et al. "Treatment with a new benzimidazole derivative bearing a pyrrolidine side chain overcomes sorafenib resistance in hepatocellular carcinoma." Sci Rep. 2019 Nov 21;9(1):17259. PMID:31754201
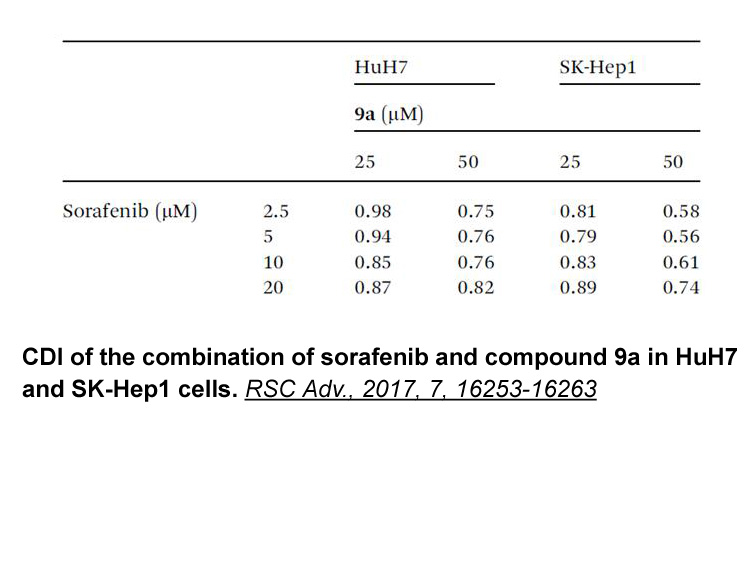
- 3. Ming-Hua Hsu, Shih-Ming Hsu, et al. "Treatment with low-dose sorafenib in combination with a novel benzimidazole derivative bearing a pyrolidine side chain provides synergistic anti-proliferative effects against human liver cancer." RSC Adv., 2017, 7, 16253-16263.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 637.03 |
| Cas No. | 475207-59-1 |
| Formula | C21H16ClF3N4O3·C7H8O3S |
| Solubility | ≥31.85 mg/mL in DMSO; insoluble in H2O; ≥4.15 mg/mL in EtOH with ultrasonic |
| Chemical Name | 4-[4-[[4-chloro-3-(trifluoromethyl)phenyl]carbamoylamino]phenoxy]-N-methylpyridine-2-carboxamide;4-methylbenzenesulfonic acid |
| SDF | Download SDF |
| Canonical SMILES | CC1=CC=C(C=C1)S(=O)(=O)O.CNC(=O)C1=NC=CC(=C1)OC2=CC=C(C=C2)NC(=O)NC3=CC(=C(C=C3)Cl)C(F)(F)F |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
| 细胞实验 [1]: | |
|
细胞系 |
MV4-11细胞和EOL-1细胞 |
|
制备方法 |
在DMSO中的溶解度大于31.9 mg/mL。若配制更高浓度的溶液,一般步骤如下:请将试管置于37 °C加热10分钟和/或将其置于超声波浴中震荡一段时间。原液于-20 °C可放置数月。 |
|
反应条件 |
100 pM ~ 10 μM;MV4-11细胞:72小时,EOL-1细胞:24小时 |
|
实验结果 |
在浓度为100 nM时,Sorafenib 促进43.6 ± 5.2%细胞经历细胞凋亡。在EOL-1细胞中,即使浓度低至10 nM,Sorafenib也能促进89.29 ± 1.8%细胞经历细胞凋亡。对经过Sorafenib治疗的MV4-11细胞的细胞周期进行分析,结果显示,Sorafenib呈剂量依赖性地诱导细胞周期停滞,处于G0/G1期的细胞百分比从52.7 ± 0.9%(对照组)增至66.8 ± 1.5%(100 nM Sorafenib组)。 |
| 动物实验 [1]: | |
|
动物模型 |
携带FLT3-ITD肿瘤的无胸腺小鼠 |
|
给药剂量 |
0.3、1.0、3或10 mg/kg;口服给药 |
|
实验结果 |
在携带FLT3-ITD肿瘤的无胸腺小鼠中,Sorafenib显示出剂量依赖性的抗肿瘤活性。在3 mg/kg和10 mg/kg的剂量下,10只小鼠中的6和9只分别显示完全的反应。MV4-11肿瘤的蛋白印迹实验结果显示,在第2次给药3小时后,STAT5磷酸化被完全抑制。同时,磷酸化组蛋白H3水平也显著降低。 |
|
注意事项 |
请于室内测试所有化合物的溶解度。虽然化合物的实际溶解度可能与其理论值略有不同,但仍处于实验系统误差的允许范围内。 |
|
References: [1]. D Auclair, D Miller, V Yatsula, et al. Antitumor activity of sorafenib in FLT3-driven leukemic cells. Leukemia, 2007, 21:439-445. |
|
| Description | Sorafenib Tosylate(Bay 43-9006)是一种多靶点激酶抑制剂,作用于Raf-1、B-Raf和VEGFR-2,IC50值分别为6 nM、22 nM和90 nM。 | |||||
| 靶点 | Raf-1 | B-Raf | VEGFR2 | PDGFRβ | ||
| IC50 | 6 nM | 22 nM | 90 nM | 57 nM | ||
质量控制和MSDS
- 批次:
化学结构
相关生物数据
相关生物数据
相关生物数据
相关生物数据
相关生物数据