Pregnenolone Carbonitrile
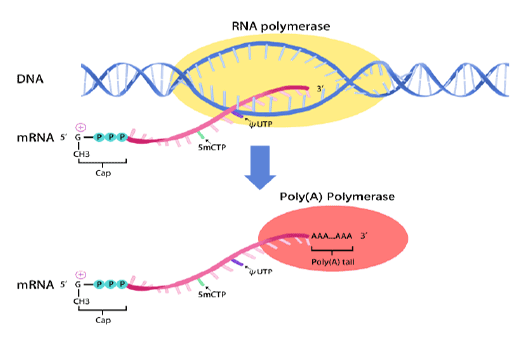
mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail
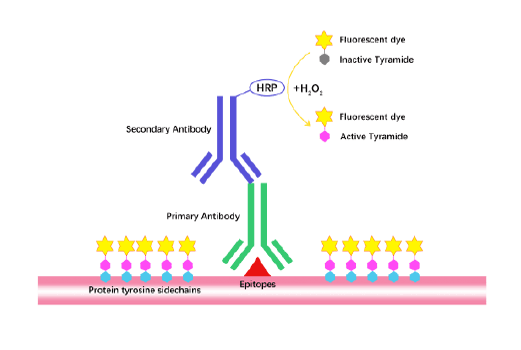
Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.
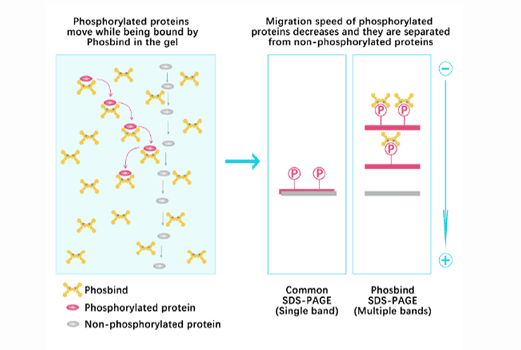
Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody
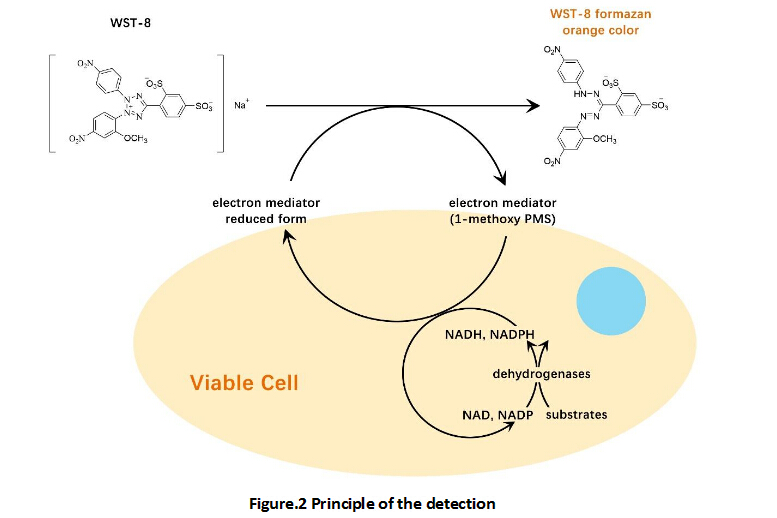
Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay
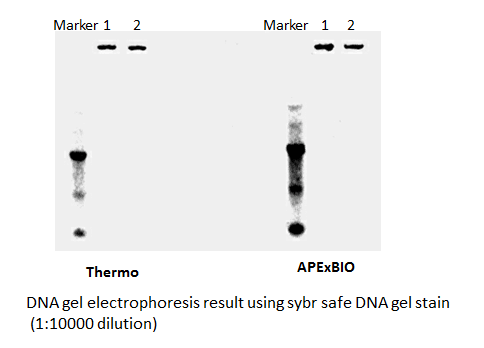
SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.
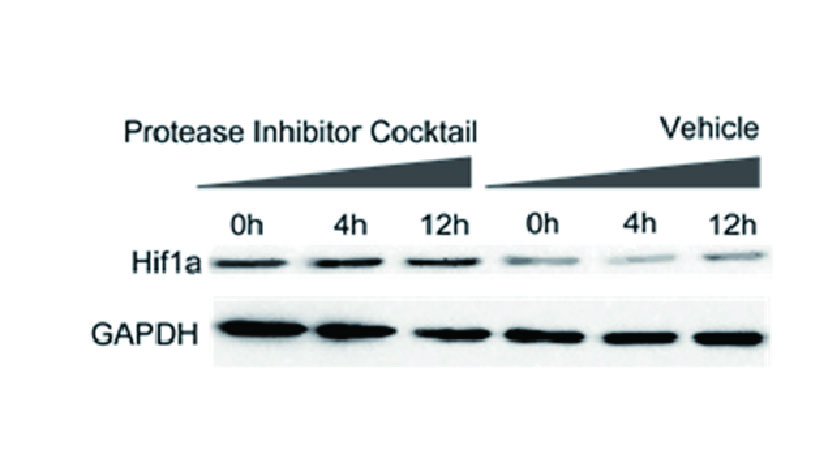
Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
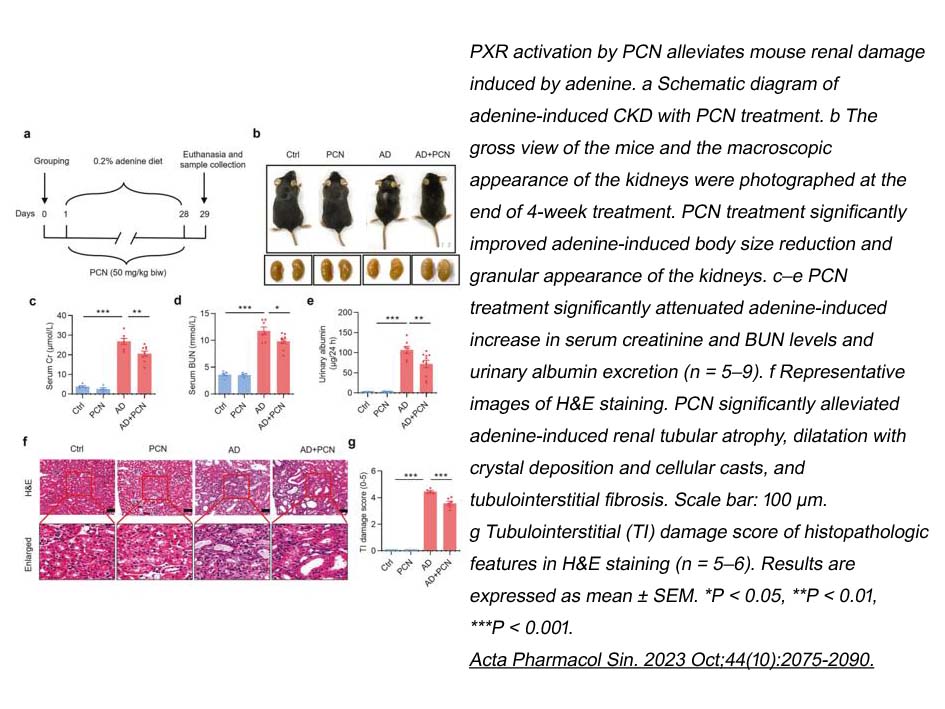
Pregnenolone-16α-carbonitrile is a rodent pregnane X receptor (PXR) activator involved in inducing the synthesis of a unique cytochrome P450 peptide in hepatic microsomes of male rats [1].
The nuclear pregnane X receptor (PXR) is an important component of the body’s adaptive defense mechanism against toxic substances. PXR could be activated by a large number of endogenous and exogenous chemicals such as antibiotics, steroids, antimycotics, and bile acids. PXR acts as a generalized sensor of hydrophobic toxins. PXR heterodimer binds with the 9-cis retinoic acid receptor (NR2B) to DNA response elements in the regulatory regions of cytochrome P450 3A monooxygenase genes and a number of other genes involved in the metabolism and elimination of xenobiotics from the body [2].
In vitro: In rat HSCs, PCN inhibited the trans-differentiation of rat HSCs in vitro despite the absence of PXR expression [3].
In vivo: PCN administration (25 mg/kg; one injection/week) to rats significantly increased the relative liver weight. In rats, PCN administration did not result in liver damage or significantly affect the level of liver damage caused by carbon tetrachloride. PCN treatment to carbon tetrachloride-treated rats resulted in a significant decrease in both intralobular α-smooth-muscle-actin immunostaining and intense Sirius Red staining in liver sections. PCN didn’t interfere with the metabolism of carbon tetrachloride to toxic metabolites [3].
References:
[1] Birnbaum L S, Baird M B, Massie H R. Pregnenolone-16alpha-carbonitrile-inducible cytochrome P450 in rat liver[J]. Research communications in chemical pathology and pharmacology, 1976, 15(3): 553.
[2] Kliewer S A, Goodwin B, Willson T M. The nuclear pregnane X receptor: a key regulator of xenobiotic metabolism[J]. Endocrine reviews, 2002, 23(5): 687-702.
[3] Marek C J, Tucker S J, Konstantinou D K, et al. Pregnenolone-16α-carbonitrile inhibits rodent liver fibrogenesis via PXR (pregnane X receptor)-dependent and PXR-independent mechanisms[J]. Biochemical Journal, 2005, 387(3): 601-608.
- 1. Wen-Hua Ming, Zhi-Lin Luan, et al. "Pregnane X receptor activation alleviates renal fibrosis in mice via interacting with p53 and inhibiting the Wnt7a/β-catenin signaling." Acta Pharmacol Sin. 2023 Oct;44(10):2075-2090. PMID: 37344564
- 2. Yingjian Chen, Tianqi Cui, et al. "Hepatic ZBTB22-mediated detoxification ameliorates acetaminophen-induced liver injury by inhibiting pregnane X receptor signaling." iScience. 2023 Mar 2;26(4):106318. PMID: 36950116
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 341.5 |
| Cas No. | 1434-54-4 |
| Formula | C22H31NO2 |
| Synonyms | PCN,Pregnenolone 16α-carbonitrile,SC-4674 |
| Solubility | insoluble in H2O; insoluble in EtOH; ≥14.17 mg/mL in DMSO |
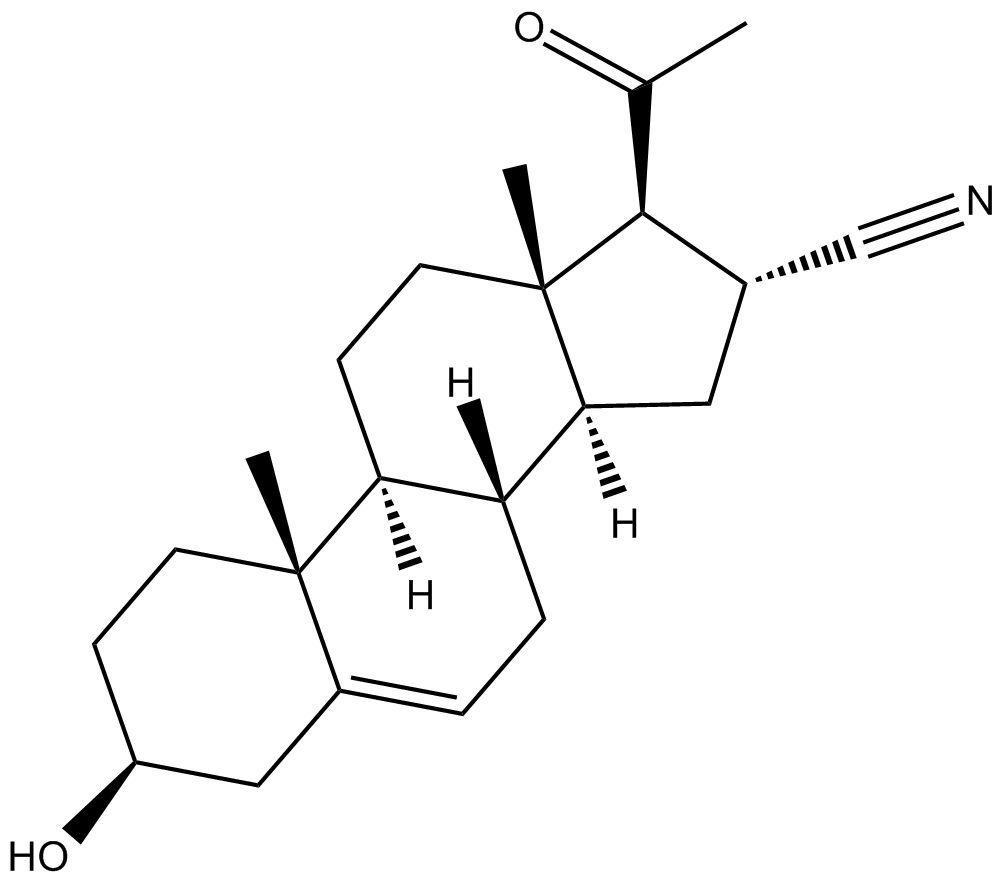
| Chemical Name | 3β-hydroxy-20-oxo-pregn-5-ene-16α-carbonitrile |
| SDF | Download SDF |
| Canonical SMILES | O[C@H](C1)CC[C@@]2(C)C1=CC[C@]3([H])[C@]2([H])CC[C@@]4(C)[C@@]3([H])C[C@@H](C#N)[C@@H]4C(C)=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构
相关生物数据