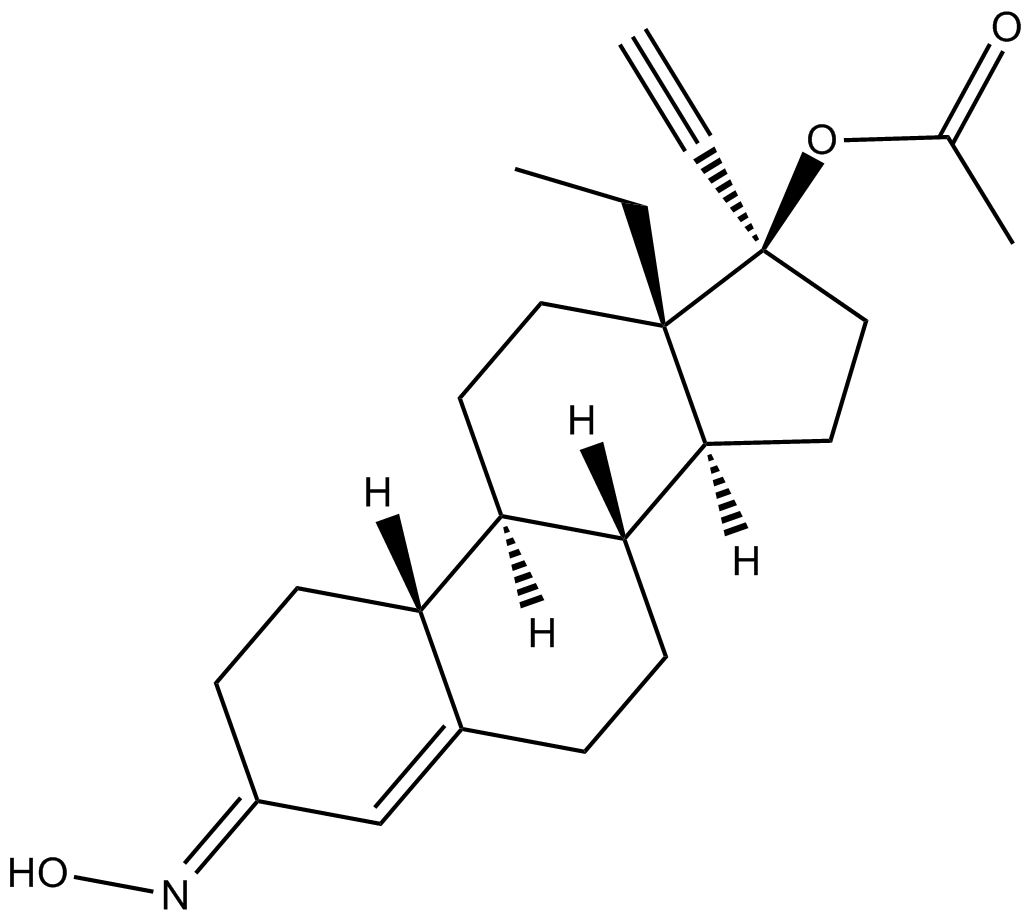
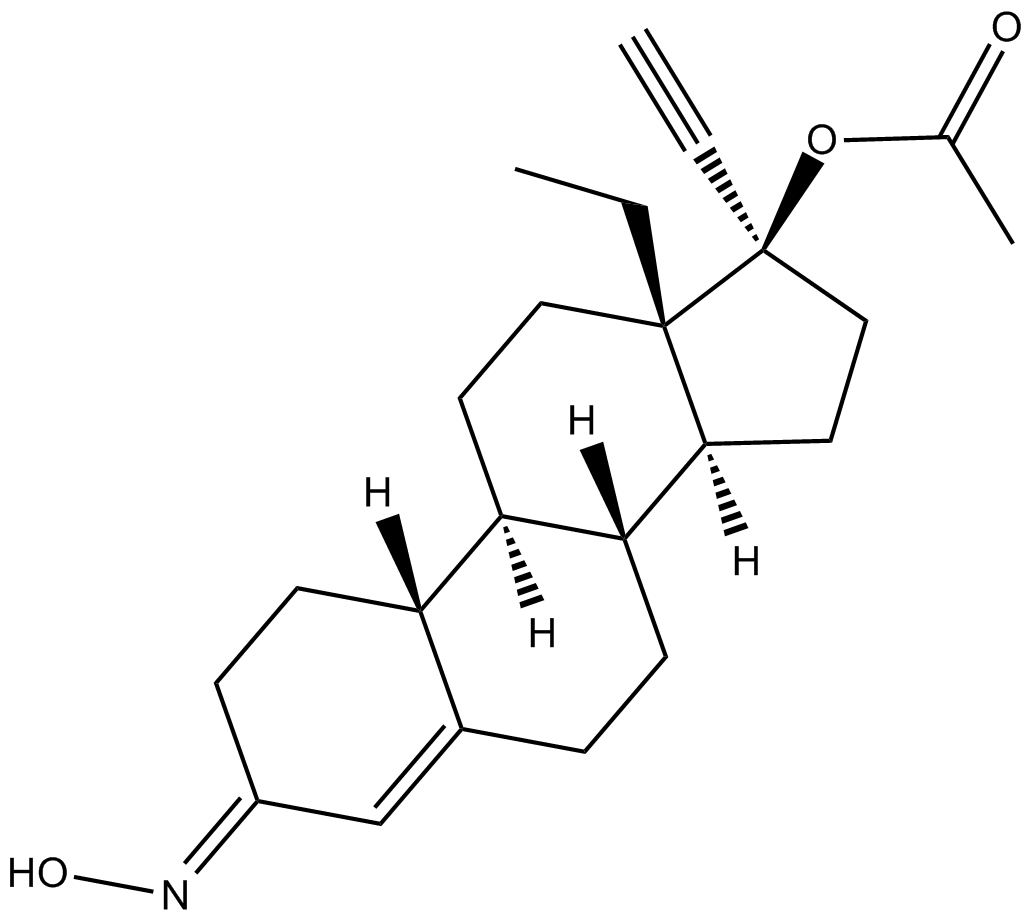
Norgestimate
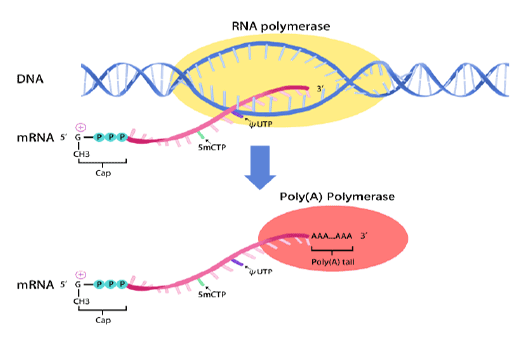
mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail
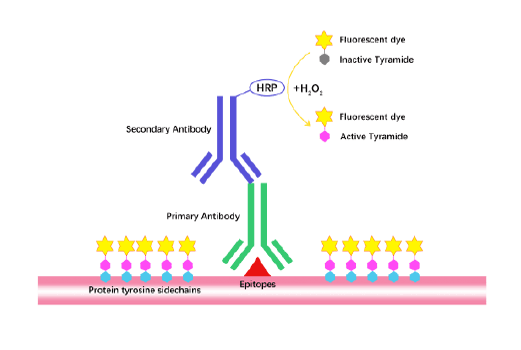
Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.
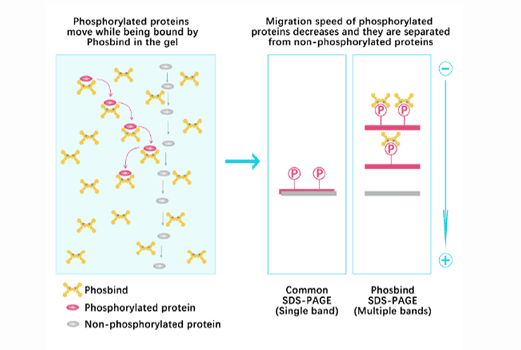
Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody
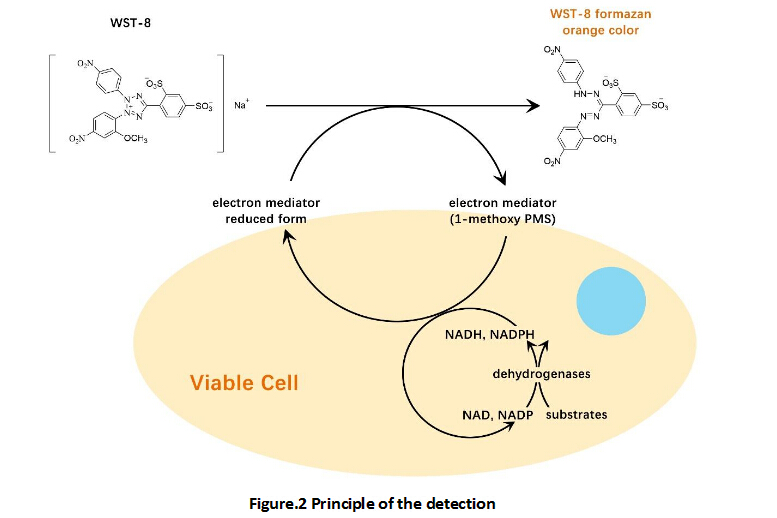
Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay
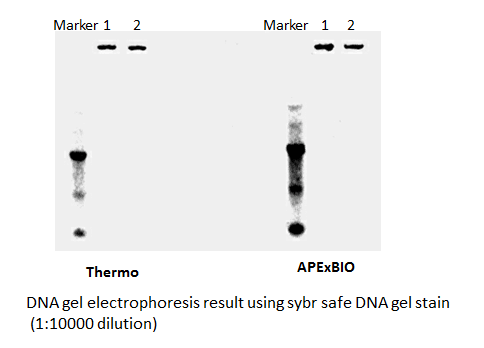
SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.
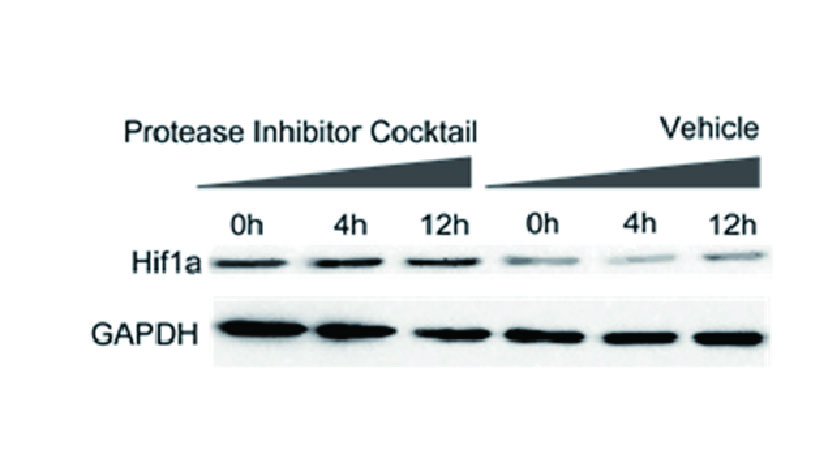
Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Norgestimate is a synthetic progesterone analog.
Progesterone, an endogenous steroid and progestogen sex hormone, is involved in the pregnancy, menstrual cycle, and embryogenesis. Progesterone is reported to belong to steroid hormones, and is the major progestogen in the body. Progesterone is also a crucial metabolic intermediate in the production of other endogenous steroids and plays an key role in brain function as a neurosteroid.
In vitro: Norgestimate was found that, unlike other 19-nortestosterone derivatives, showed high selectivity for the progesterone receptor and low androgenic activity. Moreover, norgestimate and its main active metabolite norelgestromin could not bind to or occupy sex hormone-binding globulin [1].
In vivo: The androgenic and the progestational activity of norgestimate were compared in two animal studies. It was found the difference in the pharmacological response in norgestimate treated rats was equivalent to the difference in the exposure of the animals to either directly administered or metabolically derived levonorgestrel [2].
Clinical trial: Norgestimate (brand names Ortho-Cyclen, Ortho Tri-Cyclen, Previfem, Sprintec, Prefest, others) is a steroidal progestin of the 19-nortestosterone group that is clinically used in combination with ethinylestradiol and in combination with estradiol in menopausal hormone replacement therapy [3].
References:
[1] Thomas L. Lemke; David A. Williams (2008). Foye's Principles of Medicinal Chemistry. Lippincott Williams & Wilkins. pp. 1316–. ISBN 978-0-7817-6879-5.
[2] Kuhnz W, Beier S. Comparative progestational and androgenic activity of norgestimate and levonorgestrel in the rat. Contraception. 1994 Mar;49(3):275-89.
[3] https://en. wikipedia.org/wiki/Norgestimate
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 369.5 |
| Cas No. | 35189-28-7 |
| Formula | C23H31NO3 |
| Synonyms | Dexnorgestrel acetime |
| Solubility | ≤2mg/ml in ethanol;14mg/ml in DMSO;16mg/ml in dimethyl formamide |
| Chemical Name | (17α)-17-(acetyloxy)-13-ethyl-18,19-dinorpregn-4-en-20-yn-3-one 3-oxime |
| SDF | Download SDF |
| Canonical SMILES | CC[C@@]12[C@](CC[C@@]2(OC(C)=O)C#C)([H])[C@]3([H])CCC4=C/C(CC[C@]4([H])[C@@]3([H])CC1)=N/O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构