α-Naphthoflavone
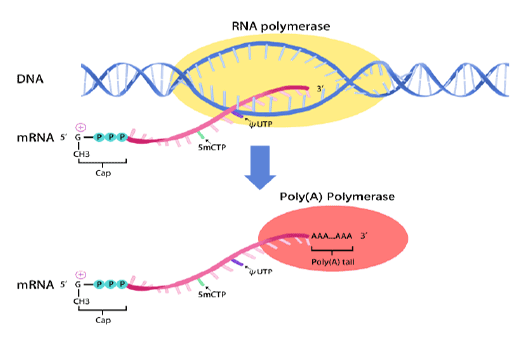
mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail
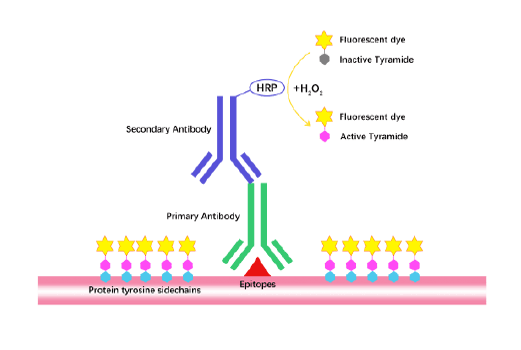
Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.
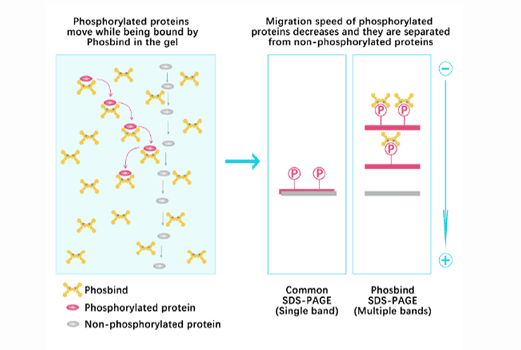
Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody
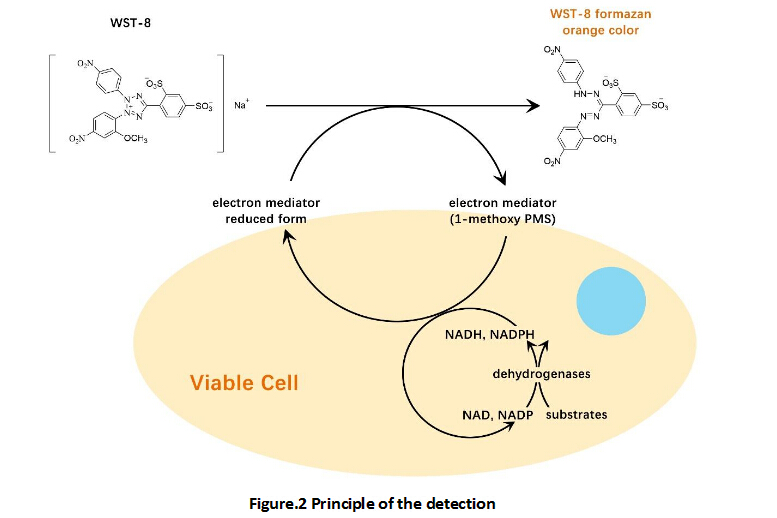
Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay
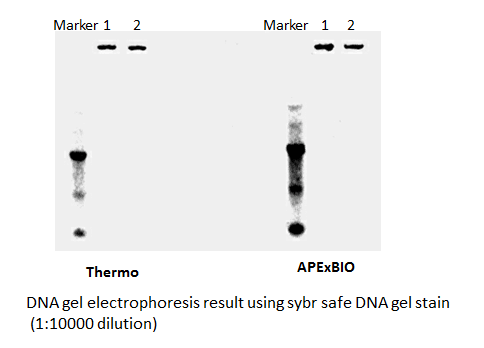
SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.
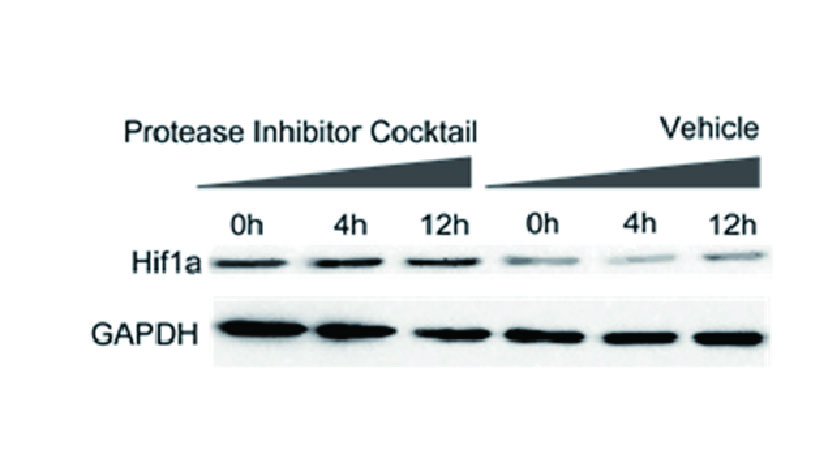
Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
α-Naphthoflavone (α-NF), also known as 7,8-benzoflavone and 2-phenyl-benzo(h)chromen-4-one, is a synthetic flavone derivative. α-Naphthoflavone is a potent inhibitor of the enzyme aromatase, which implicated in converting testosterone to estrogen [1]. It can be prepared from 2-naphthol and cinnamaldehyde [2]. α-naphthoflavone functions as both an Ah receptor antagonist and an inhibitor of cytochrome P450 activity. α-naphthoflavone stimulated P450 3A4 by selectively binding and activating an otherwise inactive subpopulation of this P450 and promoting benzo[a]pyrene binding to the latter [3].
In vitro: α-NF inhibited microsomal rabbit CYP3A6 and human CYP3A4 irreversibly. α-NF and β-NF strongly inhibited CYP1A-mediated ethoxyresorufin O-deethylase (EROD) activity with the Ki value of 9.1 ± 0.8 and 7.6 ± 1.1 nM, respectively. α-NF at 0.5, 5, 50 and 500 μM inhibited liver microsome-catalyzed AFB1-DNA binding by 22, 58, 84 and 91%, respectively [4].
In vivo: α-NF inhibited CYP1A enzymes and caused both synergism and antagonism of retene toxicity to rainbow trout (Oncorhynchus mykiss) [5]. In male Sprague–Dawley rats, treatment with αNF had no significant effects on body mass after 5 days and caused only minor increases of liver, kidney, and heart CYP1A1 mRNA. In contrast, lung CYP1A1 mRNA was increased by αNF treatment to levels comparable to that seen with β-NF treatment [6].
References:
[1]. Campbell D R, Kurzer M S. Flavonoid inhibition of aromatase enzyme activity in human preadipocytes[J]. The Journal of steroid biochemistry and molecular biology, 1993, 46(3): 381-388.
[2]. Harvey R G, HAHN J T A I, Bukowska M, et al. A new chromone and flavone synthesis and its utilization for the synthesis of potentially antitumorigenic polycyclic chromones and flavones[J]. Journal of organic chemistry, 1990, 55(25): 6161-6166.
[3]. Koley A P, Buters J T M, Robinson R C, et al. Differential mechanisms of cytochrome P450 inhibition and activation by α-naphthoflavone[J]. Journal of Biological Chemistry, 1997, 272(6): 3149-3152.
[4]. Takahashi N, Miranda C L, Henderson M C, et al. Inhibition of in vitro aflatoxin B1-DNA binding in rainbow trout by CYP1A inhibitors: α-naphthoflavone, β-naphthoflavone and trout CYP1A1 peptide antibody[J]. Comparative Biochemistry and Physiology Part C: Pharmacology, Toxicology and Endocrinology, 1995, 110(3): 273-280.
[5]. Hodson P V, Qureshi K, Noble C A J, et al. Inhibition of CYP1A enzymes by α-naphthoflavone causes both synergism and antagonism of retene toxicity to rainbow trout (Oncorhynchus mykiss)[J]. Aquatic toxicology, 2007, 81(3): 275-285.
[6]. Sinal C J, Webb C D, Bend J R. Differential in vivo effects of α‐naphthoflavone and β‐naphthoflavone on CYP1A1 and CYP2E1 in rat liver, lung, heart, and kidney[J]. Journal of biochemical and molecular toxicology, 1999, 13(1): 29-40.
| Storage | Store at -20°C |
| M.Wt | 272.3 |
| Cas No. | 604-59-1 |
| Formula | C19H12O2 |
| Synonyms | 7,8-Benzoflavone,NSC 407011 |
| Solubility | insoluble in EtOH; insoluble in H2O; ≥10 mg/mL in DMSO |
| Chemical Name | 2-phenyl-4H-naphtho[1,2-b]pyran-4-one |
| SDF | Download SDF |
| Canonical SMILES | O=C1C2=C(C(C=CC=C3)=C3C=C2)OC(C4=CC=CC=C4)=C1 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
质量控制和MSDS
- 批次:
化学结构



