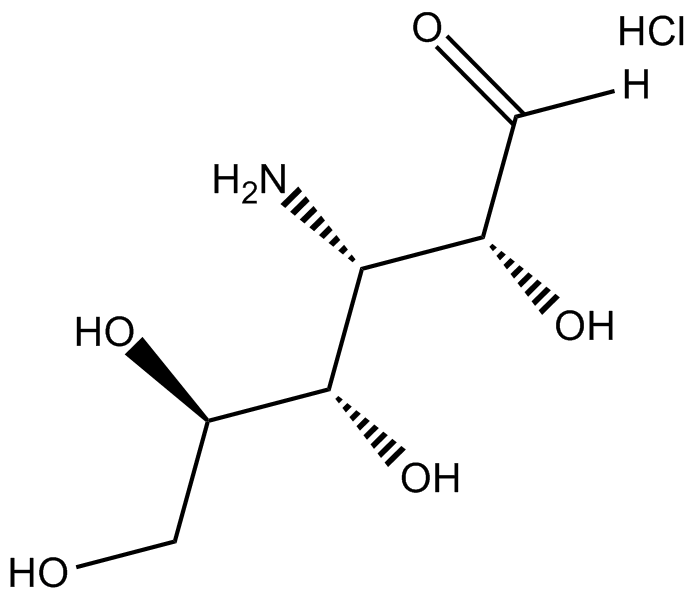
Kanosamine (hydrochloride)
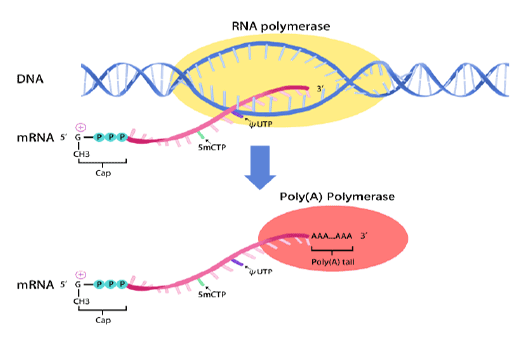
mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail
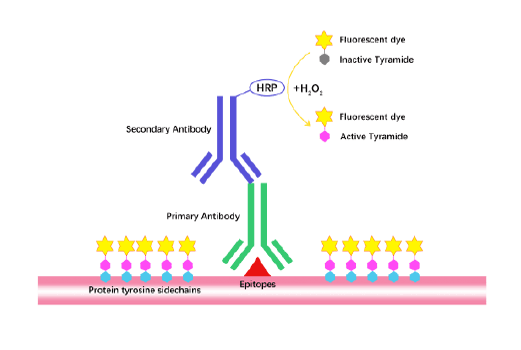
Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.
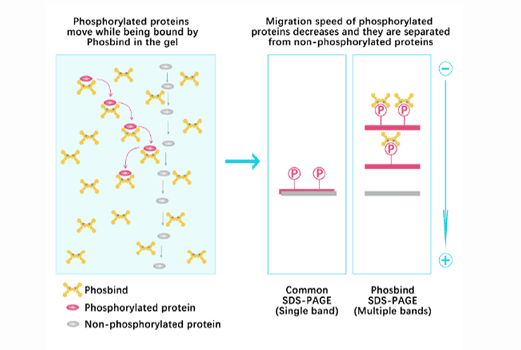
Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody
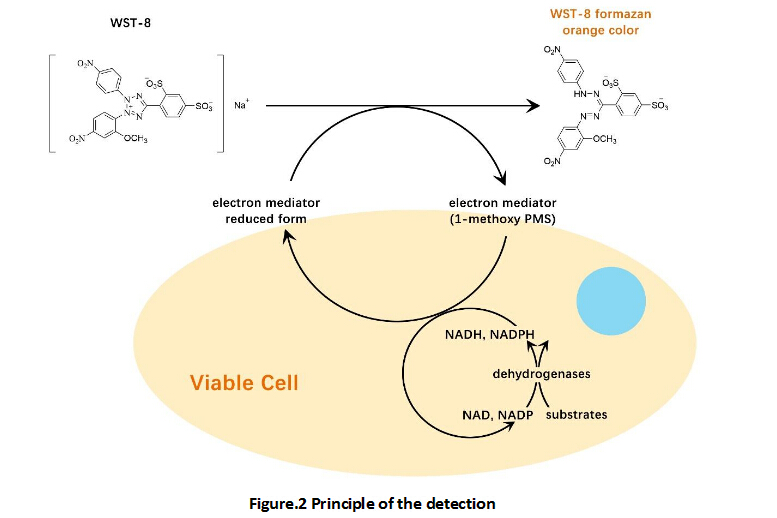
Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay
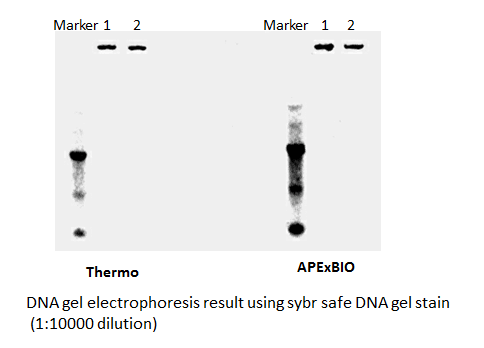
SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.
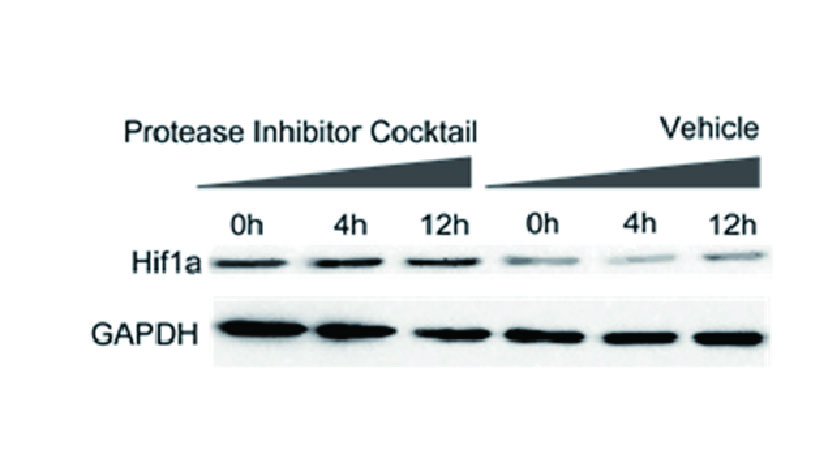
Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Kanosamine is the antibiotic produced by Bacillus cereus UW85. Kanosamine showed highly inhibitory effects to the growth of plant-pathogenic oomycetes and moderately inhibitory effects to certain fungi and inhibited few bacterial species tested. Kanosamine accumulation in B. cereus UW85 culture supernatants was enhanced by the addition of ferric iron and suppressed by addition of phosphate to rich medium. Kanosamine accumulation was also enhanced more than 300% by the addition of alfalfa seedling exudate to minimal medium [1].
Kanosamine was also produced by a Streptomyces SP [2]. Kanosamine inhibited cell wall synthesis in plant-pathogenic oomycetes with MIC value of 25 μg/ml for P. medicaginis M2913 and certain fungi as well as some bacterial species with MIC value of 400 μg/ml for S. aureus [1,2]. It has been explored as an alternative and/or supplement to synthetic pesticides and genetic resistance of crop plants for the management of plant disease [1,2].
The antibiotic kanosamine inhibited the growth of many human pathogenic fungi. Kanosamine was transported into cells by the glucose transport system and subsequently phosphorylated to generate kanosamine-6-phosphate. The product was an inhibitor of the enzyme glucosamine-6-phosphate synthase. The Inhibitory effect was competitive to one of the substrates, D-fructose-6-phosphate, with Ki value of 5.9 mM. The action of kanosamine on C. albicans cells lead to profound morphological changes, inhibition of septum formation and cell agglutination [3].
References:
[1] Milner J L, Silo-Suh L, Lee J C, et al. Production of kanosamine by Bacillus cereus UW85[J]. Applied and Environmental Microbiology, 1996, 62(8): 3061-3065.
[2] Dolak L A, Castle T M, Dietz A, et al. 3-Amino-3-deoxyglucose produced by a Streptomyces SP[J]. The Journal of antibiotics, 1980, 33(8): 900-901.
[3] Janiak A M, Milewski S. Mechanism of antifungal action of kanosamine[J]. Medical mycology, 2001, 39(5): 401-408.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 215.6 |
| Cas No. | 57649-10-2 |
| Formula | C6H13NO5·HCl |
| Solubility | ≤5mg/ml in ethanol;25mg/ml in DMSO;25mg/ml in dimethyl formamide |
| Chemical Name | 3-amino-3-deoxy-D-glucose, monohydrochloride |
| SDF | Download SDF |
| Canonical SMILES | O=C([H])[C@H](O)[C@@H](N)[C@H](O)[C@H](O)CO.Cl |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构