IL-15, human recombinant protein
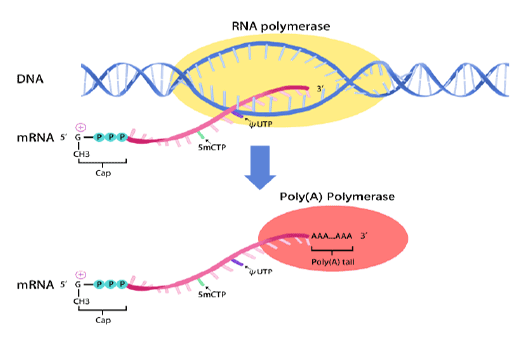
mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail
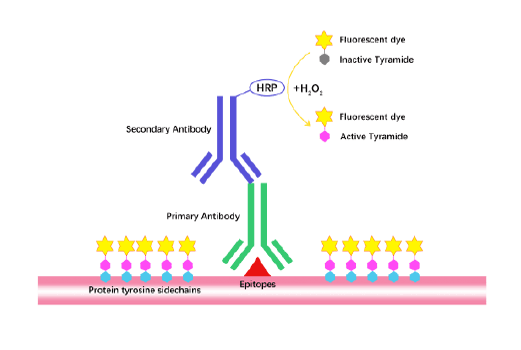
Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.
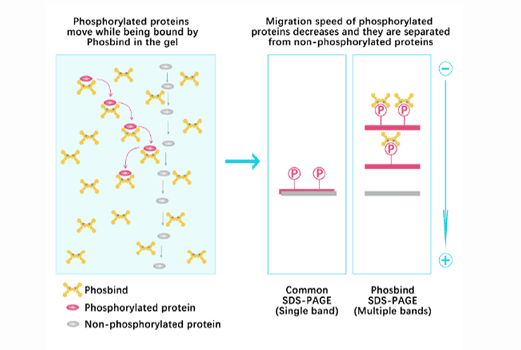
Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody
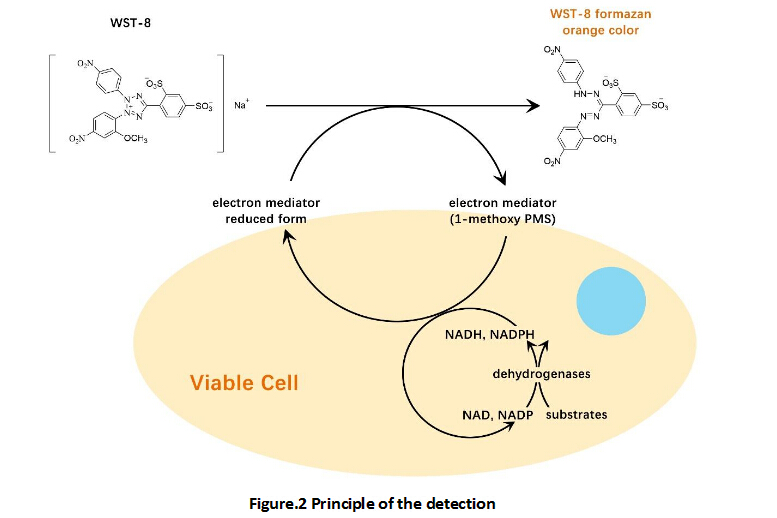
Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay
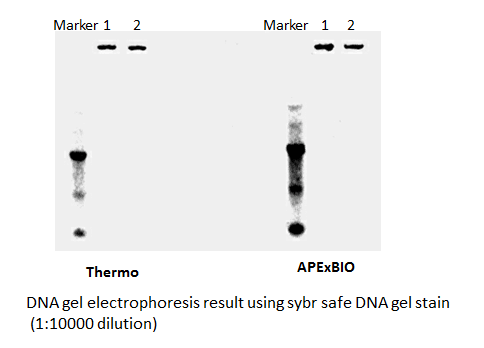
SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.
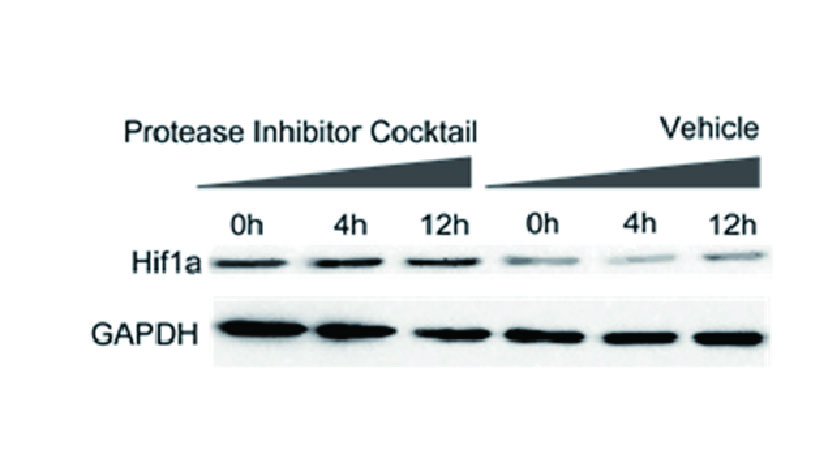
Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Human Interleukin-15 (IL-15) is expressed by the IL15 gene located on the chromosome 4. It shares approximately 97 % and 73 % sequence identity with simian and murine IL-15, respectively. Both human and simian IL-15 are active on murine cells. IL-15 is secreted by mononuclear phagocytes (and some other cells), especially macrophages following infection by virus. It possesses a variety of biological functions, including stimulating and maintaining of cellular immune responses, especially regulating T and natural killer (NK) cell activation and proliferation. In additionally, it shares many biological properties with IL-2, including T, B and NK cell-stimulatory activities. IL-15 signals through a complex composed of IL-2/IL-15 receptor beta chain. Although IL-15 lacks sequence homology with IL-2, it has recently been shown that both the beta and gamma chains of the IL-2 receptor are utilized for IL-15 binding and signaling. In addition, an IL-15 specific binding protein has also been cloned from a mouse T cell clone.
Reference:
1. Anderson DM, Johnson L, Glaccum MB, et al. 1995. Genomics, 25: 701-6
2. Krause H, Jandrig B, Wernicke C, et al. 1996. Cytokine, 8: 667-74
3. Chirifu M, Hayashi C, Nakamura T, et al. 2007. Nat Immunol, 8: 1001-7
4. Grabstein KH, Eisenman J, Shanebeck K, et al. 1994. Science, 264: 965-8
5. Giri JG, Ahdieh M, Eisenman J, et al. 1994. EMBO J, 13: 2822-30
6. Arena A, Merendino RA, Bonina L, et al. 2000. New Microbiol, 23: 105-12.
|
Gene ID |
3600 |
|
Accession # |
P40933 |
|
Alternate Names |
|
|
Source |
Escherichia coli. |
|
M.Wt |
Approximately 12.9 kDa, a single non-glycosylated polypeptide chain containing 114 amino acids. |
|
AA Sequence |
NWVNVISDLK KIEDLIQSMH IDATLYTESD VHPSCKVTAM KCFLLELQVI SLESGDASIH DTVENLIILA NNSLSSNGNV TESGCKECEE LEEKNIKEFL QSFVHIVQMF INTS |
|
Appearance |
Sterile Filtered White lyophilized (freeze-dried) powder. |
|
Stability & Storage |
Use a manual defrost freezer and avoid repeated freeze-thaw cycles. - 12 months from date of receipt, -20 to -70 °C as supplied. - 1 month, 2 to 8 °C under sterile conditions after reconstitution. - 3 months, -20 to -70 °C under sterile conditions after reconstitution. |
|
Formulation |
Lyophilized from a 0.2 µm filtered concentrated solution in PBS, pH 7.4. |
|
Reconstitution |
We recommend that this vial be briefly centrifuged prior to opening to bring the contents to the bottom. Reconstitute in sterile distilled water or aqueous buffer containing 0.1 % BSA to a concentration of 0.1-1.0 mg/mL. Stock solutions should be apportioned into working aliquots and stored at ≤ -20 °C. Further dilutions should be made in appropriate buffered solutions. |
|
Biological Activity |
Measured in a cell proliferation assay using MO7e human megakaryocytic leukemic cells. The ED50 for this effect is 0.300-2.60 ng/mL. The specific activity of recombinant human IL-15 is ≥1.50 ×108units/mg, which is calibrated against the human IL-15 reference standard (NIBSC code: 95/554). |
|
Shipping Condition |
Gel pack. |
|
Handling |
Centrifuge the vial prior to opening. |
|
Usage |
For Research Use Only! Not to be used in humans. |
Quality Control & DataSheet
- View current batch:
-
Purity: > 97 % by SDS-PAGE and HPLC analyses.
- Datasheet
Endotoxin Level: Less than 1 EU/µg of rHuIL-15 as determined by LAL method.
相关生物数据