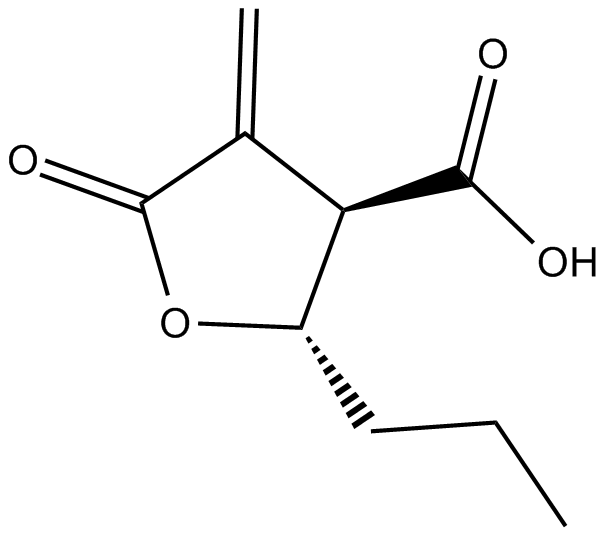
Butyrolactone 3
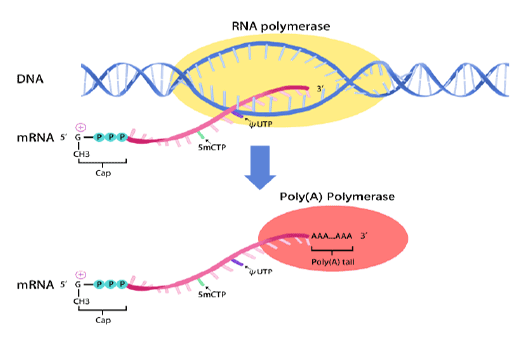
mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail
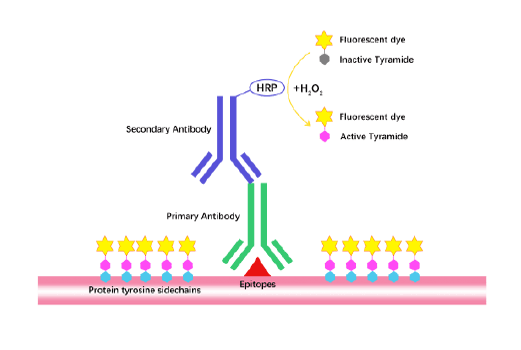
Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.
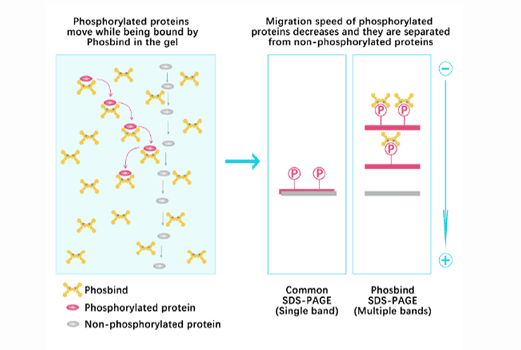
Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody
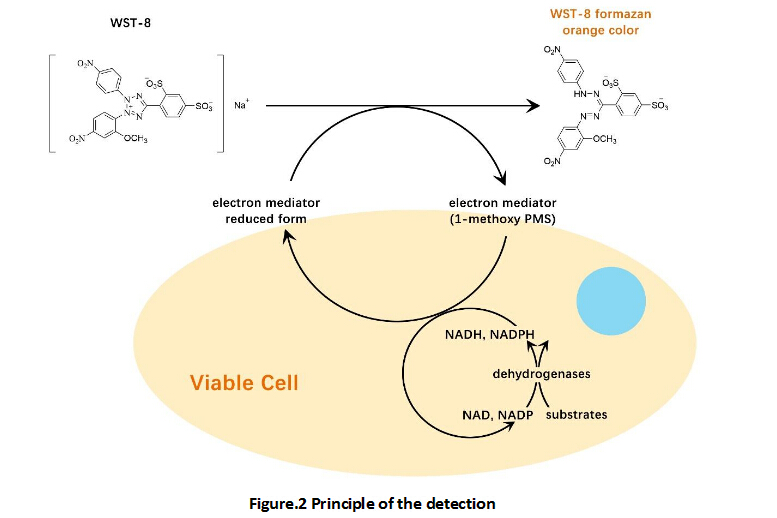
Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay
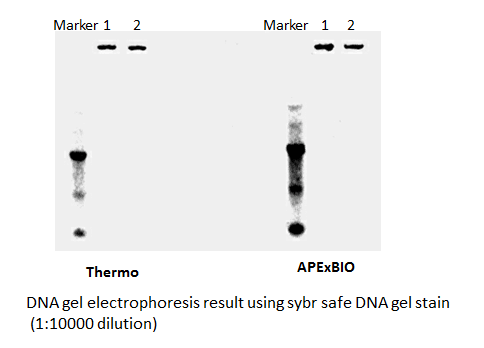
SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.
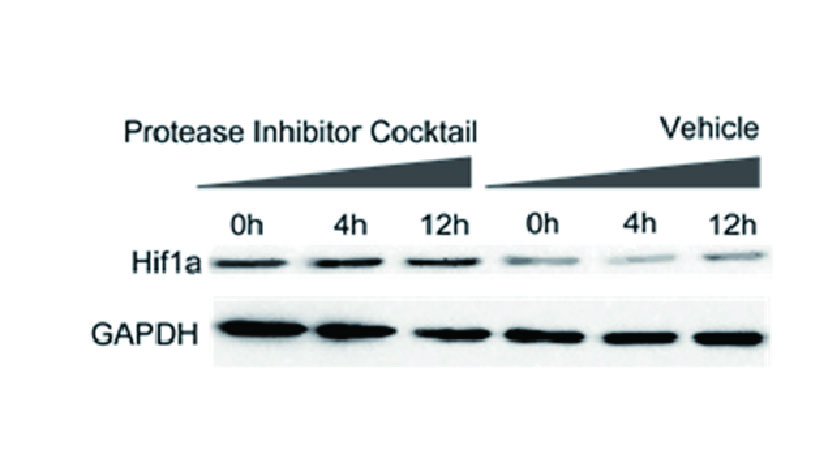
Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
IC50: 100 μM
Butyrolactone 3 is a histone acetyltransferase Gcn5 inhibitor.
The human histone acetyltransferase Gcn5 is identified as a prominent member of the GNAT family with high preference for histone H3 as a substrate.
In vitro: The analogs of butyrolactone 3 only showed a weak inhibition of CBP, while butyrolactone 3 led to an inhibition of Gcn5. It was important to see that in the presence of acetyl-CoA the Kd value for binding of histone H3 to Gcn5 or PCAF was around 100 mM. Moreover, the affinity of butyrolactone 3 to the Gcn5 enzyme was found to be comparable to that of the natural substrate H3 and could provide an good starting point for the study of SAR. In addition, a nonirreversible inhibition of Gcn5 could be determine, and thus a Michael addition of nucleophilic groups of the enzymeBs active side was unlikely. Therefore, the derivatization of gbutyrolactone 3 at position 2 might be a promising starting point for future SAR studies [1].
In vivo: So far, there is no animal in vivo data reported.
Clinical trial: Up to now, Butyrolactone 3 is still in the preclinical development stage.
Reference:
1. M. Biel, A. Kretsovali, E. Karatzali, et al. Design, synthesis, and biological evaluation of a small-molecule inhibitor of the histone acetyltransferase Gcn5. Angewandte Chemie International Edition 43, 3974-3976 (2004).
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 184.2 |
| Cas No. | 778649-18-6 |
| Formula | C9H12O4 |
| Solubility | ≤14mg/ml in ethanol;14mg/ml in DMSO;13mg/ml in dimethyl formamide |
| Chemical Name | rel-tetrahydro-4-methylene-5-oxo-2R-propyl-3S-furancarboxylic acid |
| SDF | Download SDF |
| Canonical SMILES | O=C1C([C@@H](C(O)=O)[C@H](CCC)O1)=C |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构