Amastatin (hydrochloride)
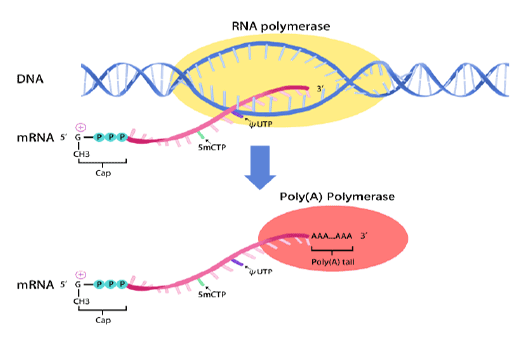
mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail
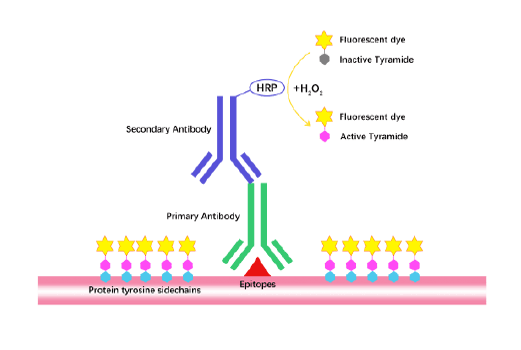
Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.
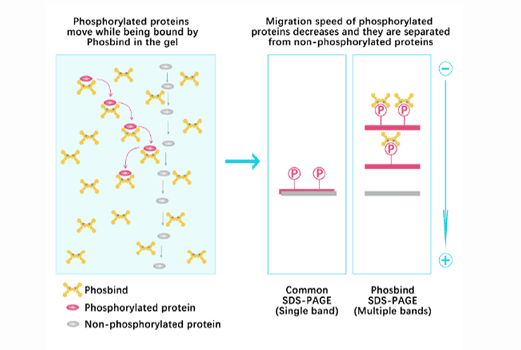
Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody
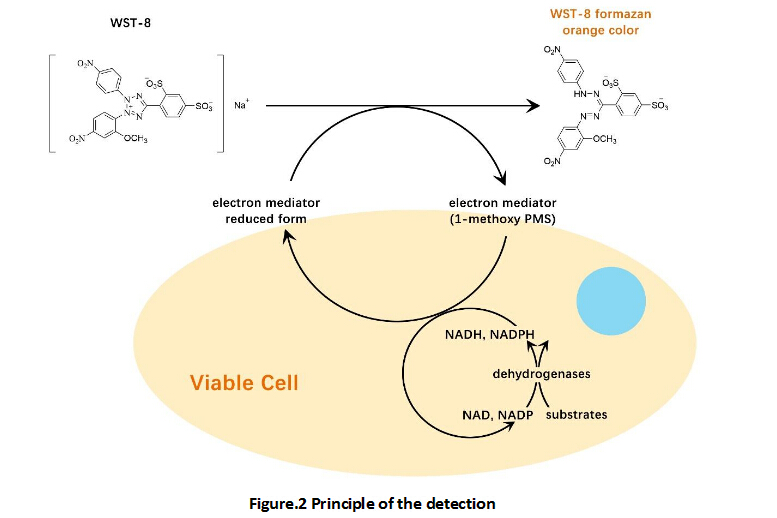
Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay
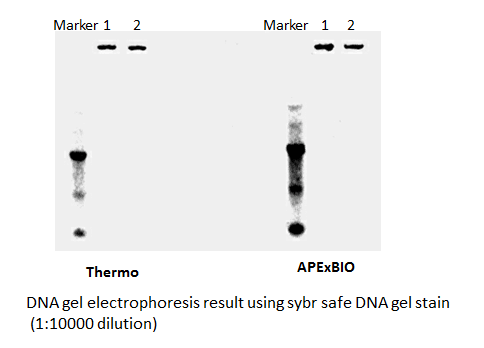
SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.
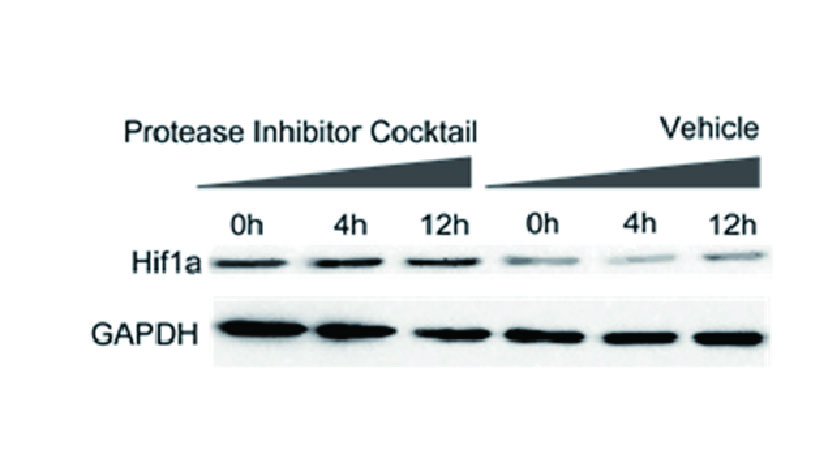
Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Amastatin (hydrochloride) is a competitive inhibitor of various aminopeptidases with slow but strong binding (Ki = 0.26, 30 and 52 nM for Aeromonas aminopeptidase, cytosolic leucine aminopeptidase and microsomal aminopeptidase) [1].
Aminopeptidases, found in many subcellular organelles, in cytosol, and as membrane components, are enzymes that catalyze the cleavage of amino acids from the amino of proteins or peptides. Aminopeptidases play important roles in various essential cellular functions [1].
Amastatin (10 μg/ml ~ 1 mg/ml) exhibited dose-dependent inhibitory profiles on E. cuniczcli replication in vitro [2]. In addition, in spleen lymphocyte cultures with macrophages, Amastatin stimulated [3H]thymidine incorporation into lymphocytes/macrophages, with the optimal stimulatory concentration around 0.1 μg/ml [3].
In anesthesized rats, infusion of amastatin (16 nmol/min i.v.) only slightly increased the potency of angiotensin I and AII, whilst significantly increased the potency of AIII and des(Asp1)angiotensin I. Furthermore, amastatin specifically inhibited plasma aminopeptidase M and aminopeptidase A activities by 81% and 10%, respectively [4].
References:
[1]. Wilkes S H, Prescott J M. The slow, tight binding of bestatin and amastatin to aminopeptidases. Journal of Biological Chemistry, 1985, 260(24): 13154-13162.
[2]. Millership J J, Didier E S, Okhuysen P C, et al. In vitro and in vivo evaluation of aminopeptidase inhibitors as antimicrosporidial therapies. Journal of Eukaryotic Microbiology, 2001, Suppl: 95S-98S.
[3]. Weissmann N, Leyhausen G, Maidhof A, et al. Mitogenic potentials of bestatin, amastatin, arphamenines A and B, FK-156 and FK-565 on spleen lymphocytes. The Journal of Antibiotics, 1985, 38(6): 772-778.
[4]. Ahmad S, Ward P E. Role of aminopeptidase activity in the regulation of the pressor activity of circulating angiotensins. Journal of Pharmacology and Experimental Therapeutics, 1990, 252(2): 643-650.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 511.01 |
| Cas No. | 100938-10-1 |
| Formula | C21H39ClN4O8 |
| Solubility | ≤1mg/ml in ethanol;2mg/ml in DMSO;10mg/ml in dimethyl formamide |
| Chemical Name | (S)-2-((S)-2-((S)-2-((2S,3R)-3-amino-2-hydroxy-5-methylhexanamido)-3-methylbutanamido)-3-methylbutanamido)succinic acid hydrochloride |
| SDF | Download SDF |
| Canonical SMILES | CC(C)[C@H](NC([C@@H](NC([C@@H](O)[C@H](N)CC(C)C)=O)C(C)C)=O)C(N[C@@H](CC(O)=O)C(O)=O)=O.Cl |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |



