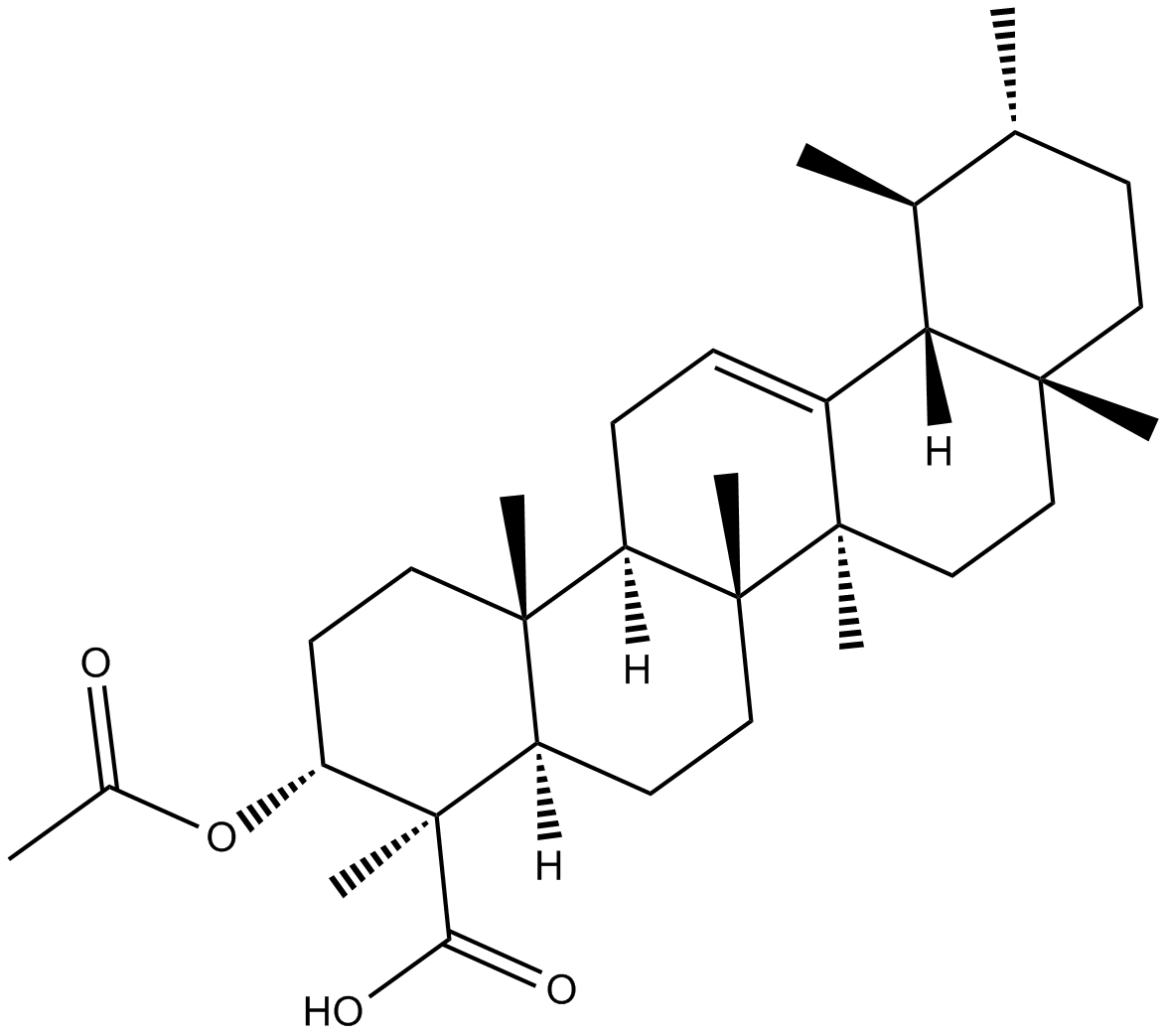
β-acetyl-Boswellic Acid
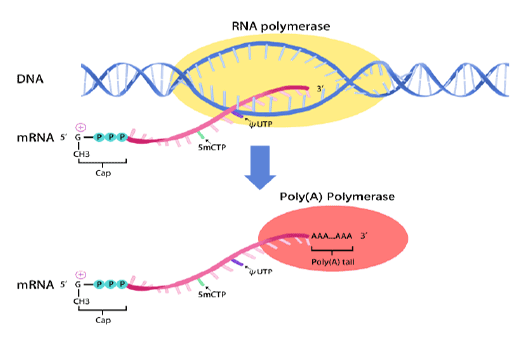
mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail
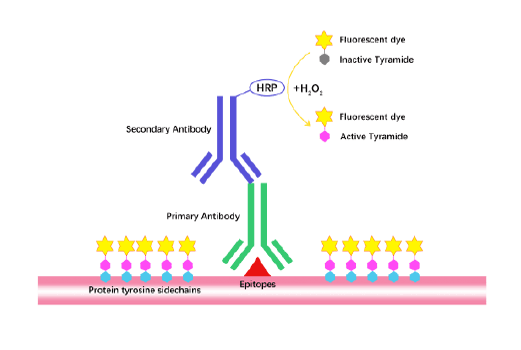
Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.
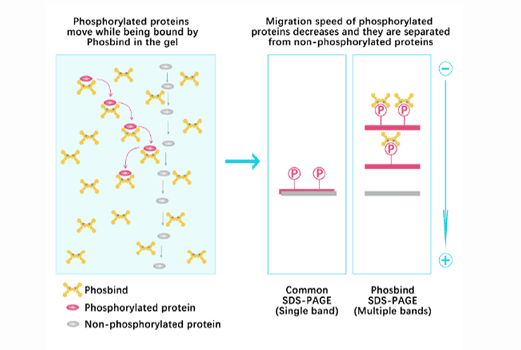
Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody
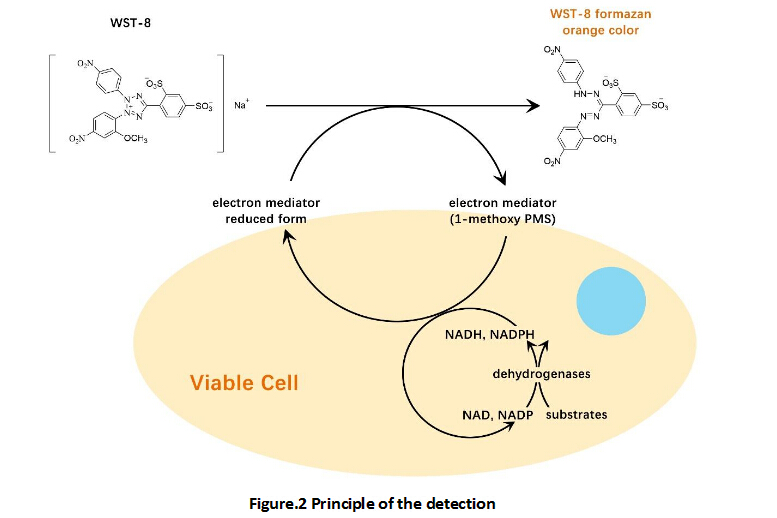
Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay
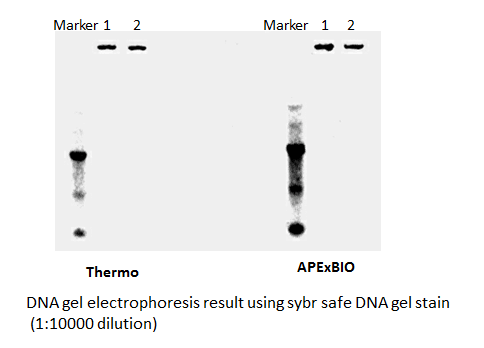
SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.
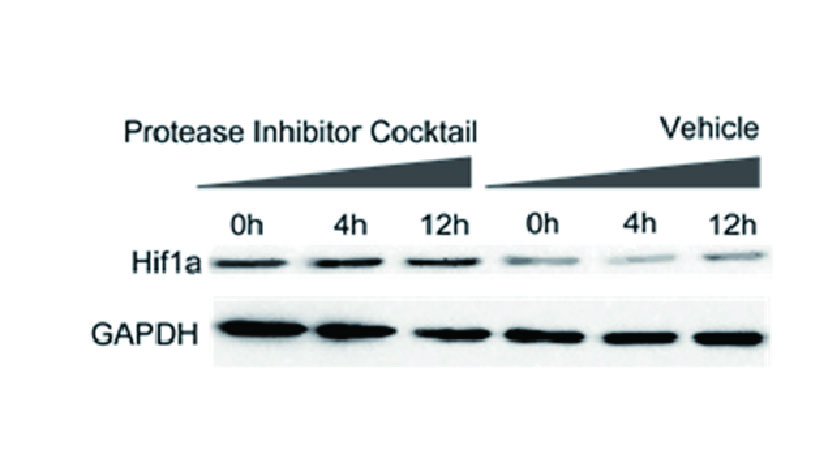
Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
β-acetyl-Boswellic Acid is a specific, nonreducing-type inhibitor of the 5-LO product formation with selectivity over 12-LO and COX [1][2].
5-lipoxygenase (5-LO) is a member of the lipoxygenase family of enzymes and transforms EFA substrates into leukotrienes.
In rat peritoneal neutrophils, boswellic acids concentration dependently decreased the formation of leukotriene B4 from endogenous arachidonic acid. In isolated human platelets, boswellic acids in concentrations up to 400 μM did not impair the cyclooxygenase and 12-lipoxygenase [1]. In calcium/calcium ionophore-stimulated peritoneal neutrophils of rats, boswellic acids significantly decreased production of LTB4 and total 5-lipoxygenase products with EC50 value of about 30 μg/ml [2]. In six human myeloid leukemia cell lines, boswellic acid acetate, a 1:1 mixture of α-boswellic acid acetate and β-boswellic acid acetate induces apoptosis through caspase-mediated pathways [4].
Boswellic acids are pentacyclic triterpenes with putative anti-inflammatory effects. In a mouse model of osteoarthritis (OA), boswellic acid significantly attenuated articular cartilage erosion and significantly reduced knee synovitis and osteophyte formation. In synovium of mice, boswellic acid directly inhibit IL-1β and TLR4 induced inflammatory cytokines production [3].
References:
[1]. Safayhi H, Mack T, Sabieraj J, et al. Boswellic acids: novel, specific, nonredox inhibitors of 5-lipoxygenase. J Pharmacol Exp Ther. 1992 Jun;261(3):1143-6.
[2]. Ammon HP, Safayhi H, Mack T, et al. Mechanism of antiinflammatory actions of curcumine and boswellic acids. J Ethnopharmacol. 1993 Mar;38(2-3):113-9.
[3]. Wang Q, Pan X, Wong HH, et al. Oral and topical boswellic acid attenuates mouse osteoarthritis. Osteoarthritis Cartilage. 2014 Jan;22(1):128-32.
[4]. Xia L, Chen D, Han R, et al. Boswellic acid acetate induces apoptosis through caspase-mediated pathways in myeloid leukemia cells. Mol Cancer Ther. 2005 Mar;4(3):381-8.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 498.7 |
| Cas No. | 5968-70-7 |
| Formula | C32H50O4 |
| Synonyms | ABA,3-O-acetyl-β-Boswellic acid,β-Boswellic acid acetate |
| Solubility | ≤5mg/ml in ethanol;25mg/ml in DMSO;25mg/ml in dimethyl formamide |
| Chemical Name | (3α,4β)-3-(acetyloxy)-urs-12-en-23-oic acid |
| SDF | Download SDF |
| Canonical SMILES | C[C@]1(C(O)=O)[C@H](OC(C)=O)CC[C@]2(C)[C@@]3([H])CC=C4[C@](CC[C@]5(C)[C@@]4([H])[C@@H](C)[C@H](C)CC5)(C)[C@]3(C)CC[C@]21[H] |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |